Restoring proton transfer in L212Q mutant of photosynthetic reaction center
Pokkuluri, P.R., Laible, P.D., Hanson, D.K., Schiffer, M.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Reaction center protein L chain | A [auth L] | 281 | Cereibacter sphaeroides | Mutation(s): 1 Gene Names: pufL Membrane Entity: Yes | 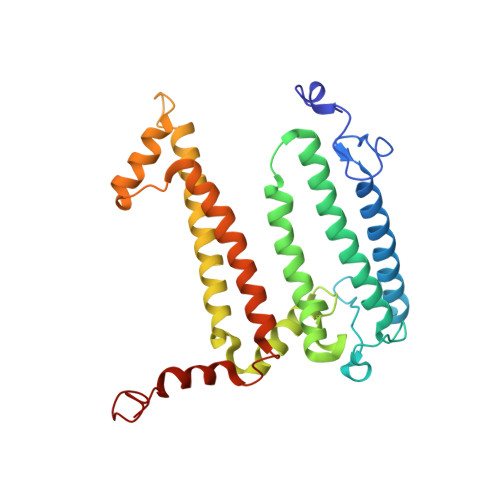 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0C0Y8 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Reaction center protein M chain | B [auth M] | 314 | Cereibacter sphaeroides | Mutation(s): 1 Gene Names: pufM Membrane Entity: Yes |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0C0Y9 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Reaction center protein H chain | C [auth H] | 260 | Cereibacter sphaeroides | Mutation(s): 0 Gene Names: puhA Membrane Entity: Yes | 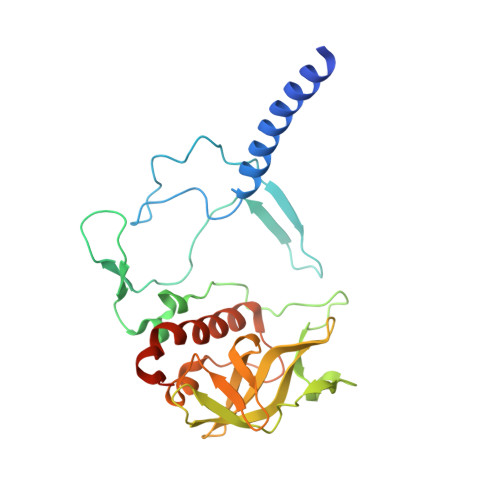 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0C0Y7 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 7 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| CDL Download:Ideal Coordinates CCD File | O [auth M] | CARDIOLIPIN C81 H156 O17 P2 XVTUQDWPJJBEHJ-KZCWQMDCSA-L |  | ||
| BCL Download:Ideal Coordinates CCD File | D [auth L], E [auth L], J [auth M], K [auth M] | BACTERIOCHLOROPHYLL A C55 H74 Mg N4 O6 DSJXIQQMORJERS-AGGZHOMASA-M |  | ||
| BPH Download:Ideal Coordinates CCD File | F [auth L], L [auth M] | BACTERIOPHEOPHYTIN A C55 H76 N4 O6 KWOZSBGNAHVCKG-SZQBJALDSA-N |  | ||
| U10 Download:Ideal Coordinates CCD File | G [auth L], M | UBIQUINONE-10 C59 H90 O4 ACTIUHUUMQJHFO-UPTCCGCDSA-N |  | ||
| SPN Download:Ideal Coordinates CCD File | N [auth M] | SPEROIDENONE C41 H70 O2 GWQAMGYOEYXWJF-YCDPMLDASA-N |  | ||
| LDA Download:Ideal Coordinates CCD File | H [auth L], P [auth M], Q [auth M], R [auth H] | LAURYL DIMETHYLAMINE-N-OXIDE C14 H31 N O SYELZBGXAIXKHU-UHFFFAOYSA-N |  | ||
| FE Download:Ideal Coordinates CCD File | I [auth M] | FE (III) ION Fe VTLYFUHAOXGGBS-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 141.5 | α = 90 |
| b = 141.5 | β = 90 |
| c = 187 | γ = 120 |
| Software Name | Purpose |
|---|---|
| CNS | refinement |
| DENZO | data reduction |
| SCALEPACK | data scaling |
| CNS | phasing |