Structure of red fluorescent protein asRFP
Gundel, S., Kachalova, G.S., Oswald, F., Fuchs, J., Bartunik, H.D., Nienhaus, G.U., Wiedenmann, J.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| GFP-like photoswitchable fluorescent protein | A [auth L], C [auth M], E [auth R], G [auth S] | 62 | Anemonia sulcata | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9GZ28 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| GFP-like photoswitchable fluorescent protein | B [auth A], D [auth B], F [auth G], H | 167 | Anemonia sulcata | Mutation(s): 1 | 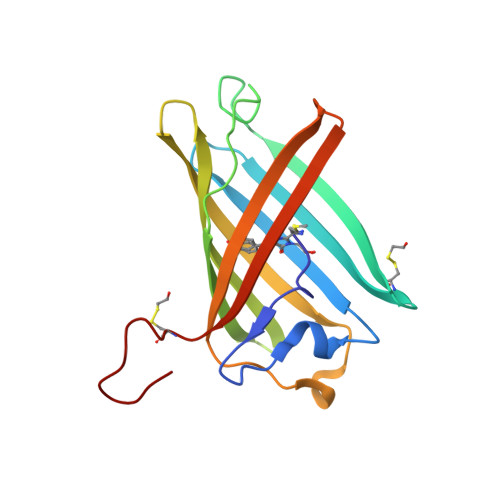 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9GZ28 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Modified Residues 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| CME Query on CME | B [auth A], D [auth B], F [auth G], H | L-PEPTIDE LINKING | C5 H11 N O3 S2 |  | CYS |
| NRQ Query on NRQ | B [auth A], D [auth B], F [auth G], H | L-PEPTIDE LINKING | C16 H17 N3 O4 S |  | MET, TYR, GLY |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 97.819 | α = 90 |
| b = 97.851 | β = 90 |
| c = 241.827 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| MAR345dtb | data collection |
| DENZO | data reduction |
| SCALEPACK | data scaling |
| MOLREP | phasing |