Crystallographic evidence of drastic movement of a copper ion toward the substrate tyrosine for starting hydroxylation reaction of tyrosinase
Matoba, Y., Yoshitsu, H., Jeon, H.J., Oda, K., Noda, M., Kumagai, T., Sugiyama, M.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Tyrosinase | 281 | Streptomyces castaneoglobisporus | Mutation(s): 0 EC: 1.14.18.1 | 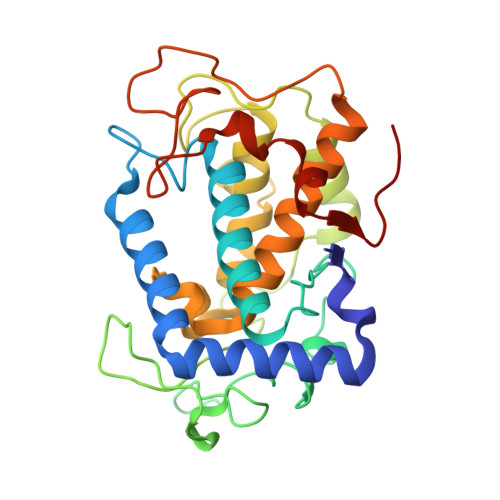 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q83WS2 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| MelC | 134 | Streptomyces castaneoglobisporus | Mutation(s): 0 | 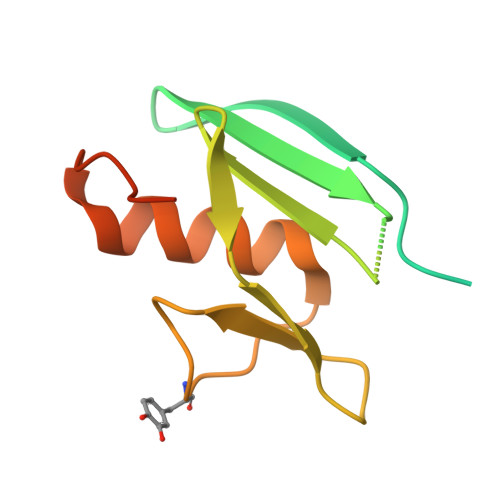 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q83WS1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| CU Download:Ideal Coordinates CCD File | C [auth A], D [auth A], E [auth A], J [auth B] | COPPER (II) ION Cu JPVYNHNXODAKFH-UHFFFAOYSA-N |  | ||
| NO3 Download:Ideal Coordinates CCD File | F [auth A], G [auth A], H [auth A], I [auth A], K [auth B] | NITRATE ION N O3 NHNBFGGVMKEFGY-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| DAH Query on DAH | B | L-PEPTIDE LINKING | C9 H11 N O4 |  | TYR |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 65.16 | α = 90 |
| b = 97.68 | β = 90 |
| c = 54.99 | γ = 90 |
| Software Name | Purpose |
|---|---|
| CNS | refinement |
| SHELXL-97 | refinement |
| BSS | data collection |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| CNS | phasing |