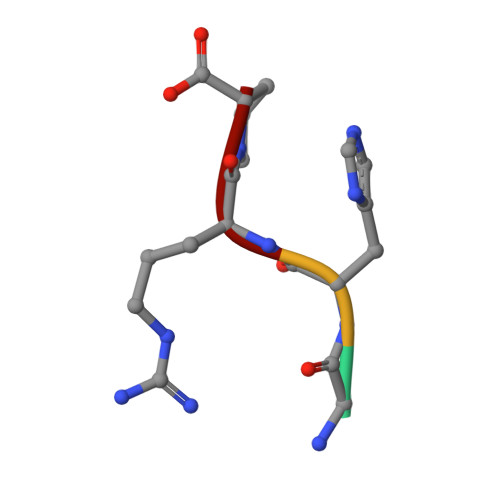
Probing the beta-chain hole of fibrinogen with synthetic peptides that differ at their amino termini
Doolittle, R.F., Pandi, L.(2007) Biochemistry 46: 10033-10038
- PubMed: 17688324
- DOI: https://doi.org/10.1021/bi7010916
- Primary Citation Related Structures:
2Q9I, 2Z4E - PubMed Abstract:
In a recent report, we showed that alanine can replace glycine at the amino terminus of synthetic B-knobs that bind to human fibrin(ogen). We now report a survey of 13 synthetic peptides with the general sequence XHRPYam, all tested with regard to their ability to delay fibrinolysis in an in vitro system activated by t-PA, the results being used as measures of binding affinity to the betaC hole. Unexpectedly, some large and bulky amino acids, including methionine and arginine, are effective binders. Amino acids that branch at the beta carbon (valine, isoleucine, and threonine) do not bind effectively. Crystal structures were determined for two of the peptides (GHRPYam and MHRPYam) complexed with fibrin fragment D-dimer; the modeling of various other side chains showed clashing in the cases of beta-carbon substituents. The two crystal structures also showed that the enhanced binding observed with pentapeptides with carboxyl-terminal tyrosine, compared with that of their tetrapeptide equivalents, is attributable to an interaction between the tyrosine side chain and a guanidino group of a nearby arginine (beta406). The equivalent position in gamma-chains of human fibrin(ogen) is occupied by a lysine (gamma338), but in chicken and lamprey fibrin(ogen), it is an arginine, just as occurs in beta chains. Accordingly, the peptides GPRPam and GPRPYam, which are surrogate A-knobs, were tested for their influence on fibrin polymerization with fibrinogen from lamprey and humans. In lampreys, GPRPYam is a significantly better inhibitor, but in humans, it is less effective than GPRPam, indicating that in the lamprey system the same tyrosine-arginine interaction can also occur in the gamma-chain setting.
- Department of Chemistry and Biochemistry and Division of Biology, University of California, San Diego, La Jolla, California 92093-0314, USA. rdoolittle@ucsd.edu
Organizational Affiliation: