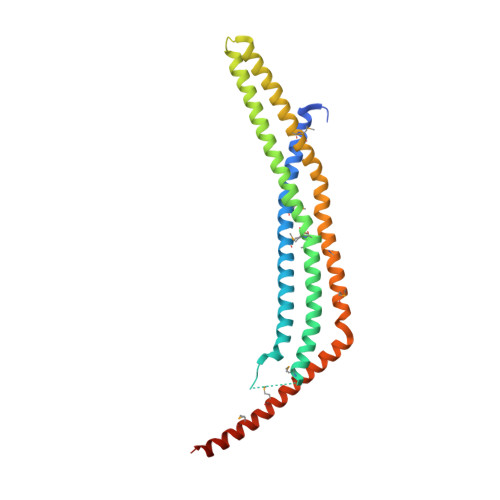
Structure of the APPL1 BAR-PH domain and characterization of its interaction with Rab5.
Zhu, G., Chen, J., Liu, J., Brunzelle, J.S., Huang, B., Wakeham, N., Terzyan, S., Li, X., Rao, Z., Li, G., Zhang, X.C.(2007) EMBO J 26: 3484-3493
- PubMed: 17581628 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/sj.emboj.7601771
- Primary Citation Related Structures:
2Q12, 2Q13 - PubMed Abstract:
APPL1 is an effector of the small GTPase Rab5. Together, they mediate a signal transduction pathway initiated by ligand binding to cell surface receptors. Interaction with Rab5 is confined to the amino (N)-terminal region of APPL1. We report the crystal structures of human APPL1 N-terminal BAR-PH domain motif. The BAR and PH domains, together with a novel linker helix, form an integrated, crescent-shaped, symmetrical dimer. This BAR-PH interaction is likely conserved in the class of BAR-PH containing proteins. Biochemical analyses indicate two independent Rab-binding sites located at the opposite ends of the dimer, where the PH domain directly interacts with Rab5 and Rab21. Besides structurally supporting the PH domain, the BAR domain also contributes to Rab binding through a small surface region in the vicinity of the PH domain. In stark contrast to the helix-dominated, Rab-binding domains previously reported, APPL1 PH domain employs beta-strands to interact with Rab5. On the Rab5 side, both switch regions are involved in the interaction. Thus we identified a new binding mode between PH domains and small GTPases.
- Crystallography Research Program, Oklahoma Medical Research Foundation, Oklahoma City, OK 73104, USA.
Organizational Affiliation: