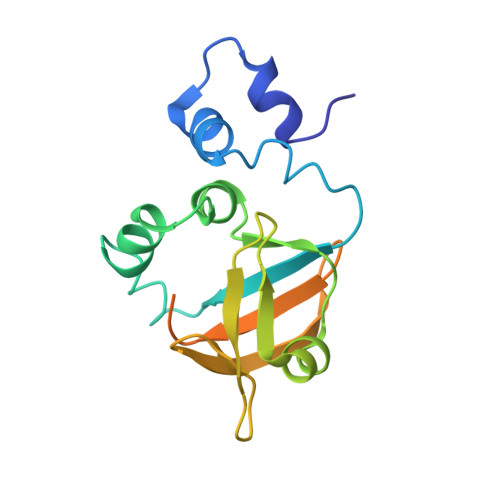
Solution structure of the NDH-1 complex subunit CupS from Thermosynechococcus elongatus.
Korste, A., Wulfhorst, H., Ikegami, T., Nowaczyk, M.M., Stoll, R.(2015) Biochim Biophys Acta 1847: 1212-1219
- PubMed: 25982357 Search on PubMed
- DOI: https://doi.org/10.1016/j.bbabio.2015.05.003
- Primary Citation Related Structures:
2MXA - PubMed Abstract:
The cyanobacterial multi-subunit membrane protein complex NDH-1 is both structurally and functionally related to Complex I of eubacteria and mitochondria. In addition to functions in respiration and cyclic electron transfer around photosystem I (PSI), the cyanobacterial NDH-1 complex is involved in a unique mechanism for inorganic carbon concentration. Although the crystal structures of the similar respiratory Complex I from Thermus thermophilus and Escherichia coli are known, atomic structural information is not available for the cyanobacterial NDH-1 complex yet. In particular, the structures of those subunits that are not homologous to Complex I will help to understand their distinct functions. The 15.7kDa protein CupS is a small soluble subunit of the complex variant NDH-1MS, which is thought to play a role in CO2 conversion. Here, we present the NMR structure of CupS from Thermosynechococcus elongatus, which is the very first structure of a specific cyanobacterial NDH-1 complex subunit. CupS shares a structural similarity with members of the Fasciclin protein superfamily. The structural comparison to Fasciclin type proteins based on known NMR structures and protein sequences of human TGFBIp, MPB70 from Mycobacterium bovis, and Fdp from Rhodobacter sphaeroides, together with a virtual docking model of CupS and NdhF3, provide first insight into the specific binding of CupS to the NDH-1MS complex at atomic resolution.
- Biomolecular Spectroscopy, Faculty of Chemistry and Biochemistry, Ruhr University of Bochum, Bochum, Germany.
Organizational Affiliation: