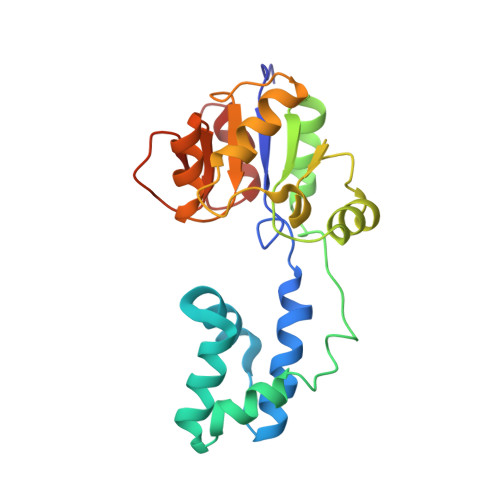
J-UNIO protocol used for NMR structure determination of the 206-residue protein NP_346487.1 from Streptococcus pneumoniae TIGR4.
Jaudzems, K., Pedrini, B., Geralt, M., Serrano, P., Wuthrich, K.(2015) J Biomol NMR 61: 65-72
- PubMed: 25428766 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1007/s10858-014-9886-3
- Primary Citation Related Structures:
2MSN, 2MU1, 2MU2 - PubMed Abstract:
The NMR structure of the 206-residue protein NP_346487.1 was determined with the J-UNIO protocol, which includes extensive automation of the structure determination. With input from three APSY-NMR experiments, UNIO-MATCH automatically yielded 77 % of the backbone assignments, which were interactively validated and extended to 97 %. With an input of the near-complete backbone assignments and three 3D heteronuclear-resolved [(1)H,(1)H]-NOESY spectra, automated side chain assignment with UNIO-ATNOS/ASCAN resulted in 77 % of the expected assignments, which was extended interactively to about 90 %. Automated NOE assignment and structure calculation with UNIO-ATNOS/CANDID in combination with CYANA was used for the structure determination of this two-domain protein. The individual domains in the NMR structure coincide closely with the crystal structure, and the NMR studies further imply that the two domains undergo restricted hinge motions relative to each other in solution. NP_346487.1 is so far the largest polypeptide chain to which the J-UNIO structure determination protocol has successfully been applied.
- Department of Integrative Structural and Computational Biology, The Scripps Research Institute, La Jolla, CA, 92037, USA.
Organizational Affiliation: