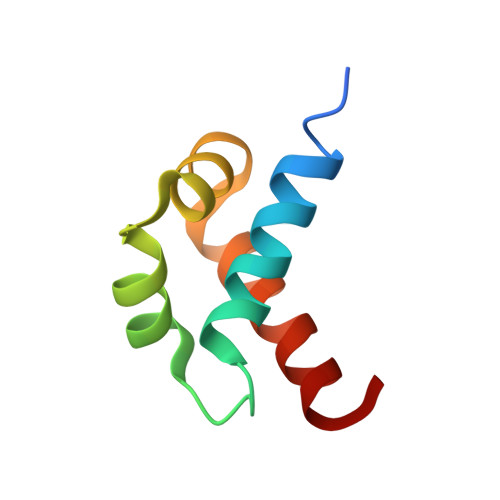
NMR structural studies on human p190-A RhoGAPFF1 revealed that domain phosphorylation by the PDGF-receptor alpha requires its previous unfolding.
Bonet, R., Ruiz, L., Aragon, E., Martin-Malpartida, P., Macias, M.J.(2009) J Mol Biology 389: 230-237
- PubMed: 19393245 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2009.04.035
- Primary Citation Related Structures:
2K85 - PubMed Abstract:
p190-A and -B Rho GAPs (guanosine triphosphatase activating proteins) are the only cytoplasmatic proteins containing FF domains. In p190-A Rho GAP, the region containing the FF domains has been implicated in binding to the transcription factor TFII-I. Moreover, phosphorylation of Tyr308 within the first FF domain inhibits this interaction. Because the structural determinants governing this mechanism remain unknown, we sought to solve the structure of the first FF domain of p190-A Rho GAP (RhoGAPFF1) and to study the potential impact of phosphorylation on the structure. We found that RhoGAPFF1 does not fold with the typical (alpha1-alpha2-3(10)-alpha 3) arrangement of other FF domains. Instead, the NMR data obtained at 285 K show an alpha1-alpha2-alpha 3-alpha 4 topology. In addition, we observed that specific contacts between residues in the first loop and the fourth helix are indispensable for the correct folding and stability of this domain. The structure also revealed that Tyr308 contributes to the domain hydrophobic core. Furthermore, the residues that compose the target motif of the platelet-derived growth factor receptor alpha kinase form part of the alpha 3 helix. We observed that the phosphorylation reaction requires a previous step including domain unfolding, a process that occurs at 310 K. In the absence of phosphorylation, the temperature-dependent RhoGAPFF1 folding/unfolding process is reversible. However, phosphorylation causes an irreversible destabilization of the RhoGAPFF1 structure, which probably accounts for the inhibitory effect that it exerts on the TFII-I interaction. Our results link the ability of a protein domain to be phosphorylated with conformational changes in its three-dimensional structure.
- The Institute for Research in Biomedicine, Barcelona (Protein NMR Group), Passeig Lluis Companys 23, Baldiri Reixac 10-13, E-08028 Barcelona, Spain.
Organizational Affiliation: