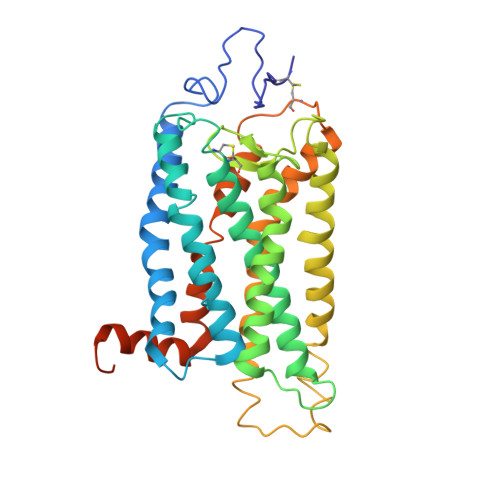
Crystal Structure of a Thermally Stable Rhodopsin Mutant.
Standfuss, J., Xie, G., Edwards, P.C., Burghammer, M., Oprian, D.D., Schertler, G.F.X.(2007) J Mol Biology 372: 1179
- PubMed: 17825322 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jmb.2007.03.007
- Primary Citation Related Structures:
2J4Y - PubMed Abstract:
We determined the structure of the rhodopsin mutant N2C/D282C expressed in mammalian cells; the first structure of a recombinantly produced G protein-coupled receptor (GPCR). The mutant was designed to form a disulfide bond between the N terminus and loop E3, which allows handling of opsin in detergent solution and increases thermal stability of rhodopsin by 10 deg.C. It allowed us to crystallize a fully deglycosylated rhodopsin (N2C/N15D/D282C). N15 mutations are normally misfolding and cause retinitis pigmentosa in humans. Microcrystallographic techniques and a 5 microm X-ray beam were used to collect data along a single needle measuring 5 microm x 5 microm x 90 microm. The disulfide introduces only minor changes but fixes the N-terminal cap over the beta-sheet lid covering the ligand-binding site, a likely explanation for the increased stability. This work allows structural investigation of rhodopsin mutants and shows the problems encountered during structure determination of GPCRs and other mammalian membrane proteins.
- MRC Laboratory of Molecular Biology, Structural Studies, Hills Road, Cambridge CB2 2QH, UK.
Organizational Affiliation: