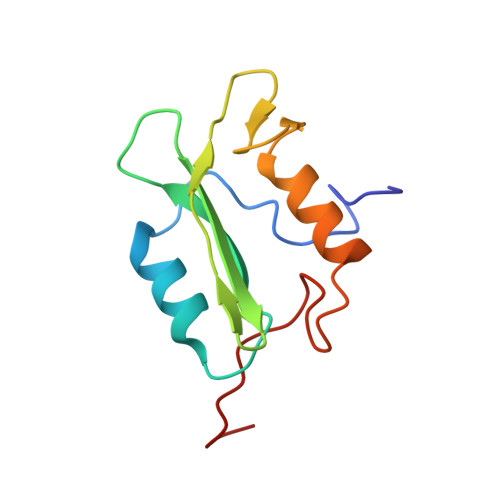
Structural basis for the requirement of two phosphotyrosine residues in signaling mediated by syk tyrosine kinase
Groesch, T.D., Zhou, F., Mattila, S., Geahlen, R.L., Post, C.B.(2006) J Mol Biology 356: 1222-1236
- PubMed: 16410013 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2005.11.095
- Primary Citation Related Structures:
2FCI - PubMed Abstract:
The protein-tyrosine kinase Syk couples immune recognition receptors to multiple signal transduction pathways, including the mobilization of calcium and the activation of NFAT. The ability of Syk to regulate signaling is influenced by its phosphorylation on tyrosine residues within the linker B region. The phosphorylation of both Y342 and Y346 is necessary for optimal signaling from the B cell receptor for antigen. The SH2 domains of multiple signaling proteins share the ability to bind this doubly phosphorylated site. The NMR structure of the C-terminal SH2 domain of PLCgamma (PLCC) bound to a doubly phosphorylated Syk peptide reveals a novel mode of phosphotyrosine recognition. PLCC undergoes extensive conformational changes upon binding to form a second phosphotyrosine-binding pocket in which pY346 is largely desolvated and stabilized through electrostatic interactions. The formation of the second binding pocket is distinct from other modes of phosphotyrosine recognition in SH2-protein association. The dependence of signaling on simultaneous phosphorylation of these two tyrosine residues offers a new mechanism to fine-tune the cellular response to external stimulation.
- Department of Medicinal Chemistry and Molecular Pharmacology, Purdue Cancer Center and Markey Center for Structural Biology, Purdue University, West Lafayette, IN 47907, USA.
Organizational Affiliation: