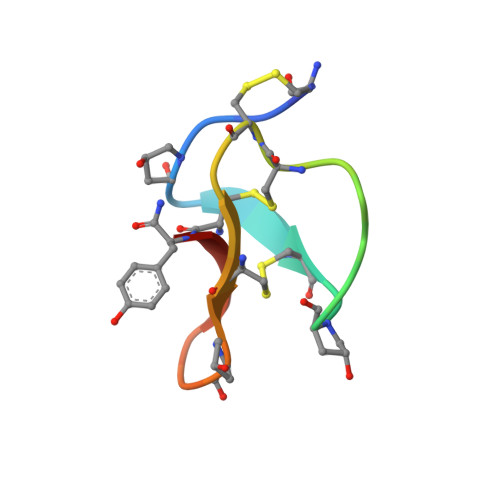
Refined solution structure of omega-conotoxin GVIA: implications for calcium channel binding
Pallaghy, P.K., Norton, R.S.(1999) J Pept Res 53: 343-351
- PubMed: 10231724 Search on PubMed
- DOI: https://doi.org/10.1034/j.1399-3011.1999.00040.x
- Primary Citation Related Structures:
2CCO - PubMed Abstract:
The polypeptide omega-conotoxin GVIA (GVIA) is an N-type calcium channel blocker from the venom of Conus geographus, a fish-hunting cone shell. Here we describe a high-resolution solution structure of this member of the 'inhibitor cystine knot' protein family. The structure, based on NMR data acquired at 600 MHz, has mean pairwise RMS differences of 0.25 +/- 0.06 and 1.07 +/- 0.14 A over the backbone heavy atoms and all heavy atoms, respectively. The solvent-accessible side chains are better defined than in previously published structures and provide an improved basis for docking GVIA with models of the calcium channel. Moreover, some side chain interactions important in GVIA folding in vitro and in stabilizing the native structure are defined clearly in the refined structure. Two qualitatively different backbone conformations in the segment from Thr11 to Asn14 persisted in the restrained simulated annealing calculations until a small number of lower bound constraints was included to prevent close contacts from occurring that did not correspond with peaks in the NOESY spectrum. It is possible that GVIA is genuinely flexible at this segment, spending a finite time in the alternative conformation, and this may influence its interaction with the calcium channel.
- Biomolecular Research Institute, Parkville, Vic., Australia.
Organizational Affiliation: