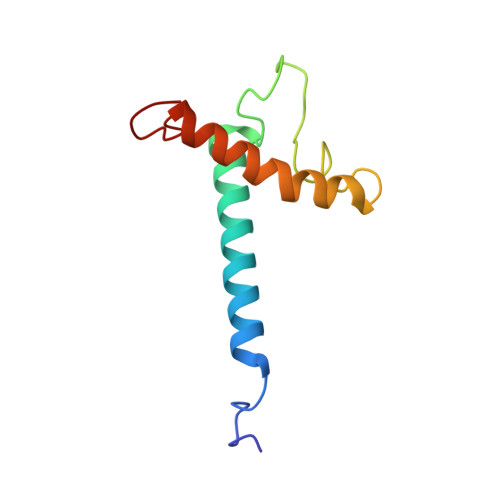
Structure, dynamics, and membrane topology of stannin: a mediator of neuronal cell apoptosis induced by trimethyltin chloride.
Buck-Koehntop, B.A., Mascioni, A., Buffy, J.J., Veglia, G.(2005) J Mol Biology 354: 652-665
- PubMed: 16246365 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2005.09.038
- Primary Citation Related Structures:
1ZZA - PubMed Abstract:
Organotin compounds or alkyltins are ubiquitous environmental toxins that have been implicated in cellular death. Unlike other xenobiotic compounds, such as organomercurials and organoleads, alkyltins activate apoptotic cascades at low concentrations. Trimethyltin (TMT) chloride is amongst the most toxic organotin compounds, and is known to selectively inflict injury to specific regions of the brain. Stannin (SNN), an 88-residue mitochondrial membrane protein, has been identified as the specific marker for neuronal cell apoptosis induced by TMT intoxication. This high specificity of TMT makes SNN an ideal model system for understanding the mechanism of organotin neurotoxicity at a molecular level. Here, we report the three-dimensional structure and dynamics of SNN in detergent micelles, and its topological orientation in lipid bilayers as determined by solution and solid-state NMR spectroscopy. We found that SNN is a monotopic membrane protein composed of three domains: a single transmembrane helix (residues 10-33) that transverses the lipid bilayer at approximately a 20 degrees angle with respect to the membrane normal; a 28 residue unstructured linker, which includes a conserved CXC metal-binding motif and a putative 14-3-3zeta binding domain; and a distorted cytoplasmic helix (residues 61-79) that is partially absorbed into the plane of the lipid bilayer with a tilt angle of approximately 80 degrees from the membrane normal. The structure and architecture of SNN within the lipid environment provides insight about how this protein transmits toxic insults caused by TMT across the membrane.
- Department of Chemistry, University of Minnesota, 207 Pleasant St. SE, Minneapolis, MN 55455-0431, USA.
Organizational Affiliation: