Structure of the unphosphorylated C-terminal tail segment of the SRC kinase and its role in SRC activity regulation
Fleury, D., Sarubbi, E., Courjaud, A., Guitton, J.D., Ducruix, A.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Proto-oncogene tyrosine-protein kinase Src | 276 | Homo sapiens | Mutation(s): 2 EC: 2.7.1.112 (PDB Primary Data), 2.7.10.2 (UniProt) | 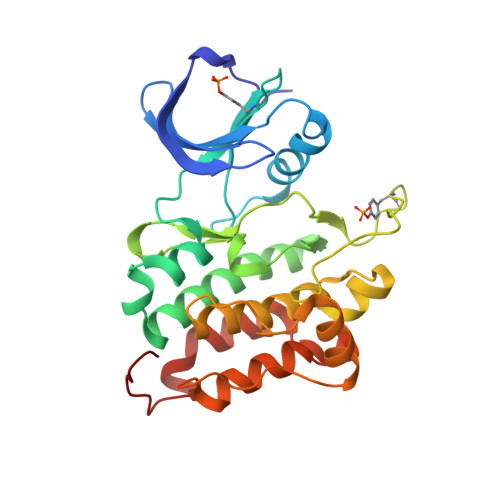 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P12931 GTEx: ENSG00000197122 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P12931 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| PTR Query on PTR | A, B | L-PEPTIDE LINKING | C9 H12 N O6 P |  | TYR |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 66.985 | α = 90 |
| b = 63.421 | β = 109.54 |
| c = 76.693 | γ = 90 |
| Software Name | Purpose |
|---|---|
| DENZO | data reduction |
| SCALA | data scaling |
| AMoRE | phasing |
| CNS | refinement |
| CCP4 | data scaling |