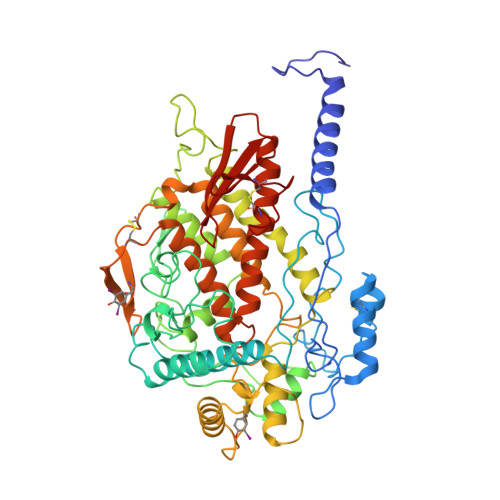
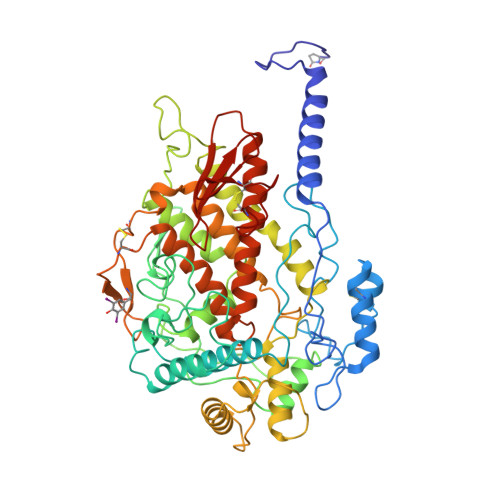
X-ray structure determination of a vanadium-dependent haloperoxidase from Ascophyllum nodosum at 2.0 A resolution.
Weyand, M., Hecht, H., Kiess, M., Liaud, M., Vilter, H., Schomburg, D.(1999) J Mol Biology 293: 595-611
- PubMed: 10543953 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.1999.3179
- Primary Citation Related Structures:
1QI9 - PubMed Abstract:
The homo-dimeric structure of a vanadium-dependent haloperoxidase (V-BPO) from the brown alga Ascophyllum nodosum (EC 1.1.11.X) has been solved by single isomorphous replacement anomalous scattering (SIRAS) X-ray crystallography at 2.0 A resolution (PDB accession code 1QI9), using two heavy-atom datasets of a tungstate derivative measured at two different wavelengths. The protein sequence (SwissProt entry code P81701) of V-BPO was established by combining results from protein and DNA sequencing, and electron density interpretation. The enzyme has nearly an all-helical structure, with two four-helix bundles and only three small beta-sheets. The holoenzyme contains trigonal-bipyramidal coordinated vanadium atoms at its two active centres. Structural similarity to the only other structurally characterized vanadium-dependent chloroperoxidase (V-CPO) from Curvularia inaequalis exists in the vicinity of the active site and to a lesser extent in the central four-helix bundle. Despite the low sequence and structural similarity between V-BPO and V-CPO, the vanadium binding centres are highly conserved on the N-terminal side of an alpha-helix and include the proposed catalytic histidine residue (His418(V-BPO)/His404(V-CPO)). The V-BPO structure contains, in addition, a second histidine near the active site (His411(V-BPO)), which can alter the redox potential of the catalytically active VO2-O2 species by protonation/deprotonation reactions. Specific binding sites for the organic substrates, like indoles and monochlordimedone, or for halide ions are not visible in the V-BPO structure. A reaction mechanism for the enzymatic oxidation of halides is discussed, based on the present structural, spectroscopic and biochemical knowledge of vanadium-dependent haloperoxidases, explaining the observed enzymatic differences between both enzymes.
- Department of Molecular Structure Research, GBF (Gesellschaft für Biotechnologische Forschung), Mascheroder Weg 1, Braunschweig, D-38124, Germany. michael-wayand@mpi-dortmund.mpg.de
Organizational Affiliation: