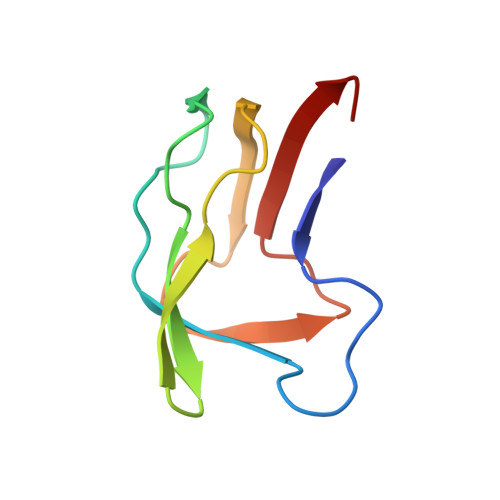
Three-dimensional structure of the lipoyl domain from Bacillus stearothermophilus pyruvate dehydrogenase multienzyme complex.
Dardel, F., Davis, A.L., Laue, E.D., Perham, R.N.(1993) J Mol Biology 229: 1037-1048
- PubMed: 8445635 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.1993.1103
- Primary Citation Related Structures:
1LAB, 1LAC - PubMed Abstract:
The structure of the lipoyl domain from the pyruvate dehydrogenase multienzyme complex of Bacillus stearothermophilus has been determined by means of nuclear magnetic resonance spectroscopy. A total of 452 nuclear Overhauser effect distance constraints and 76 dihedral angle restraints were employed as the input for the structure calculations, which were performed using a hybrid distance geometry-simulated annealing strategy and the programs DISGEO and X-PLOR. The overall structure of the lipoyl domain (residues 1 to 79 of the dihydrolipoamide acetyltransferase polypeptide chain) is that of a flattened eight-stranded beta-barrel folded around a core of well-defined hydrophobic residues. The lipoylation site, lysine 42, is located in the middle of a beta-turn, and the N and C-terminal residues of the domain are close together in adjacent beta-strands at the opposite end of the molecule. The polypeptide backbone exhibits a 2-fold axis of quasi-symmetry, with the C alpha atoms of residues 15 to 39 and 52 to 76 being almost superimposable on those of residues 52 to 76 and 15 to 39, respectively (root-mean-square deviation = 1.48 A). The amino acid residues at key positions in the structure are conserved among all the reported primary structures of lipoyl domains, suggesting that the domains all fold in a similar way.
- Laboratoire de Biochimie, URA 240 du CNRS, Ecole Polytechnique, Palaiseau, France.
Organizational Affiliation: