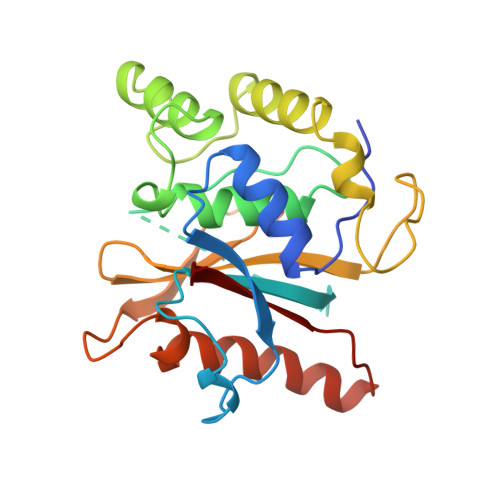
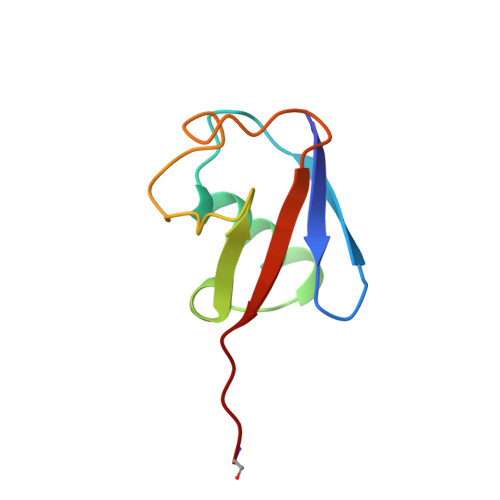
Structural basis for the specificity of ubiquitin C-terminal hydrolases.
Johnston, S.C., Riddle, S.M., Cohen, R.E., Hill, C.P.(1999) EMBO J 18: 3877-3887
- PubMed: 10406793 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/emboj/18.14.3877
- Primary Citation Related Structures:
1CMX - PubMed Abstract:
The release of ubiquitin from attachment to other proteins and adducts is critical for ubiquitin biosynthesis, proteasomal degradation and other cellular processes. De-ubiquitination is accomplished in part by members of the UCH (ubiquitin C-terminal hydrolase) family of enzymes. We have determined the 2.25 A resolution crystal structure of the yeast UCH, Yuh1, in a complex with the inhibitor ubiquitin aldehyde (Ubal). The structure mimics the tetrahedral intermediate in the reaction pathway and explains the very high enzyme specificity. Comparison with a related, unliganded UCH structure indicates that ubiquitin binding is coupled to rearrangements which block the active-site cleft in the absence of authentic substrate. Remarkably, a 21-residue loop that becomes ordered upon binding Ubal lies directly over the active site. Efficiently processed substrates apparently pass through this loop, and constraints on the loop conformation probably function to control UCH specificity.
- Department of Biochemistry, University of Iowa, Iowa City, IA 52242, USA.
Organizational Affiliation: