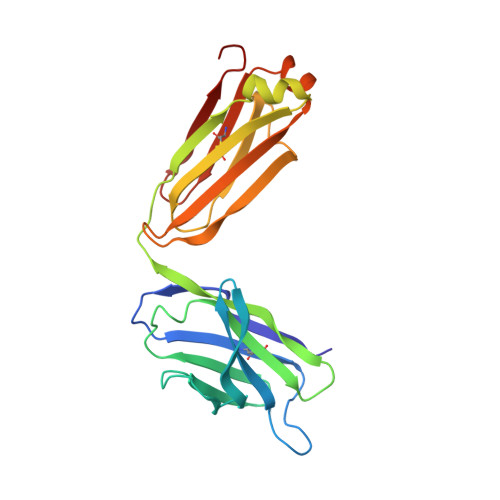
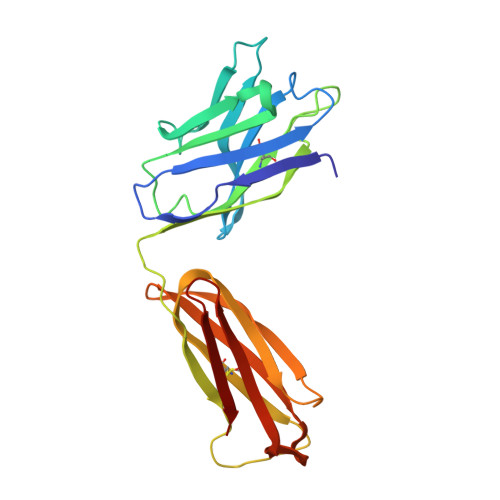
An autoantibody to single-stranded DNA: comparison of the three-dimensional structures of the unliganded Fab and a deoxynucleotide-Fab complex.
Herron, J.N., He, X.M., Ballard, D.W., Blier, P.R., Pace, P.E., Bothwell, A.L., Voss Jr., E.W., Edmundson, A.B.(1991) Proteins 11: 159-175
- PubMed: 1749770 Search on PubMed
- DOI: https://doi.org/10.1002/prot.340110302
- Primary Citation Related Structures:
1CBV, 1NBV - PubMed Abstract:
Crystal structures of the Fabs from an autoantibody (BV04-01) with specificity for single-stranded DNA have been determined in the presence and absence of a trinucleotide of deoxythymidylic acid, d(pT)3. Formation of the ligand-protein complex was accompanied by small adjustments in the orientations of the variable (VL and VH) domains. In addition, there were local conformational changes in the first hypervariable loop of the light chain and the third hypervariable loop of the heavy chain, which together with the domain shifts led to an improvement in the complementarity of nucleotide and Fab. The sugar-phosphate chain adopted an extended and "open" conformation, with the base, sugar, and phosphate components available for interactions with the protein. Nucleotide 1 (5'-end) was associated exclusively with the heavy chain, nucleotide 2 was shared by both heavy and light chains, and nucleotide 3 was bound by the light chain. The orientation of phosphate 1 was stabilized by hydrogen bonds with serine H52a and asparagine H53. Phosphate 2 formed an ion pair with arginine H52, but no other charge-charge interactions were observed. Insertion of the side chain of histidine L27d between nucleotides 2 and 3 resulted in a bend in the sugar-phosphate chain. The most dominant contacts with the protein involved the central thymine base, which was immobilized by cooperative stacking and hydrogen bonding interactions. This base was intercalated between a tryptophan ring (no. H100a) from the heavy chain and a tyrosine ring (no. L32) from the light chain. The resulting orientation of thymine was favorable for the simultaneous formation of two hydrogen bonds with the backbone carbonyl oxygen and the side chain hydroxyl group of serine L91 (the thymine atoms were the hydrogen on nitrogen 3 and keto oxygen 4).
- Department of Pharmaceutics, University of Utah, Salt Lake City 84108.
Organizational Affiliation: