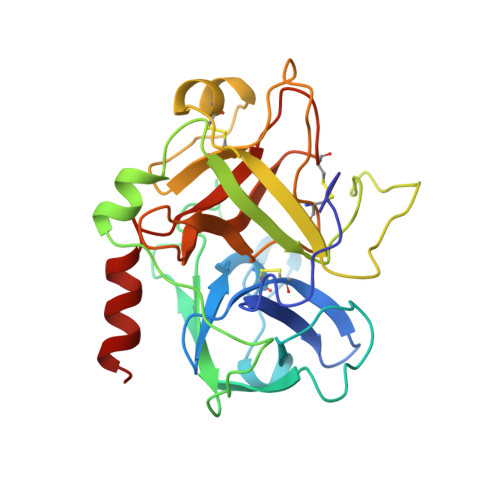
Novel natural product 5,5-trans-lactone inhibitors of human alpha-thrombin: mechanism of action and structural studies.
Weir, M.P., Bethell, S.S., Cleasby, A., Campbell, C.J., Dennis, R.J., Dix, C.J., Finch, H., Jhoti, H., Mooney, C.J., Patel, S., Tang, C.M., Ward, M., Wonacott, A.J., Wharton, C.W.(1998) Biochemistry 37: 6645-6657
- PubMed: 9578548 Search on PubMed
- DOI: https://doi.org/10.1021/bi972499o
- Primary Citation Related Structures:
1AWF, 1AWH - PubMed Abstract:
High-throughput screening of methanolic extracts from the leaves of the plant Lantana camara identified potent inhibitors of human alpha-thrombin, which were shown to be 5,5-trans-fused cyclic lactone euphane triterpenes [O'Neill et al. (1998) J. Nat. Prod. (submitted for publication)]. Proflavin displacement studies showed the inhibitors to bind at the active site of alpha-thrombin and alpha-chymotrypsin. Kinetic analysis of alpha-thrombin showed tight-binding reversible competitive inhibition by both compounds, named GR133487 and GR133686, with respective kon values at pH 8.4 of 1.7 x 10(6) s-1 M-1 and 4.6 x 10(6) s-1 M-1. Electrospray ionization mass spectrometry of thrombin/inhibitor complexes showed the tight-bound species to be covalently attached, suggesting acyl-enzyme formation by reaction of the active-site Ser195 with the trans-lactone carbonyl. X-ray crystal structures of alpha-thrombin/GR133686 (3.0 A resolution) and alpha-thrombin/GR133487 (2.2 A resolution) complexes showed continuous electron density between Ser195 and the ring-opened lactone carbonyl, demonstrating acyl-enzyme formation. Turnover of inhibitor by alpha-thrombin was negligible and mass spectrometry of isolated complexes showed that reversal of inhibition occurs by reformation of the trans-lactone from the acyl-enzyme. The catalytic triad appears undisrupted and the inhibitor carbonyl occupies the oxyanion hole, suggesting the observed lack of turnover is due to exclusion of water for deacylation. The acyl-enzyme inhibitor hydroxyl is properly positioned for nucleophilic attack on the ester carbonyl and therefore relactonization; furthermore, the higher resolution structure of alpha-thrombin/GR133487 shows this hydroxyl to be effectively superimposable with the recently proposed deacylating water for peptide substrate hydrolysis [Wilmouth, R. C., et al. (1997) Nat. Struct.Biol. 4, 456-462], suggesting the alpha-thrombin/GR133487 complex may be a good model for this reaction.
- Glaxo Wellcome Medicines Research Centre, Stevenage, Hertfordshire, U.K.
Organizational Affiliation: