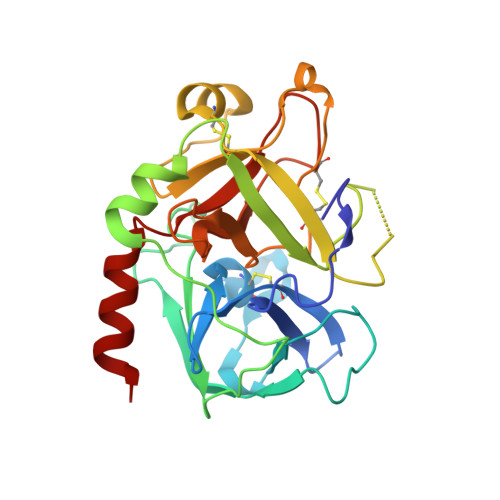
Molecular basis of thrombomodulin activation of slow thrombin
Adams, T.E., Li, W., Huntington, J.A.(2009) J Thromb Haemost 7: 1688-1695
- PubMed: 19656282
- DOI: https://doi.org/10.1111/j.1538-7836.2009.03563.x
- Primary Citation of Related Structures:
3GIS - PubMed Abstract:
Coagulation is a highly regulated process where the ability to prevent blood loss after injury is balanced against the maintenance of blood fluidity. Thrombin is at the center of this balancing act. It is the critical enzyme for producing and stabilizing a clot, but when complexed with thrombomodulin (TM) it is converted to a powerful anticoagulant. Another cofactor that may play a role in determining thrombin function is the monovalent cation Na(+). Its apparent affinity suggests that half of the thrombin generated is in a Na(+)-free 'slow' state and half is in a Na(+)-coordinated 'fast' state. While slow thrombin is a poor procoagulant enzyme, when complexed to TM it is an effective anticoagulant. To better understand this molecular transformation we solved a 2.4 A structure of thrombin complexed with EGF domains 4-6 of TM in the absence of Na(+) and other cofactors or inhibitors. We find that TM binds as previously observed, and that the thrombin component resembles structures of the fast form. The Na(+) binding loop is observed in a conformation identical to the Na(+)-bound form, with conserved water molecules compensating for the missing ion. Using the fluorescent probe p-aminobenzamidine we show that activation of slow thrombin by TM principally involves the opening of the primary specificity pocket. These data show that TM binding alters the conformation of thrombin in a similar manner as Na(+) coordination, resulting in an ordering of the Na(+) binding loop and an opening of the adjacent S1 pocket. We conclude that other, more subtle subsite changes are unlikely to influence thrombin specificity toward macromolecular substrates.
Organizational Affiliation:
Department of Haematology, Cambridge Institute for Medical Research, University of Cambridge, Cambridge CB2 0XY, UK.