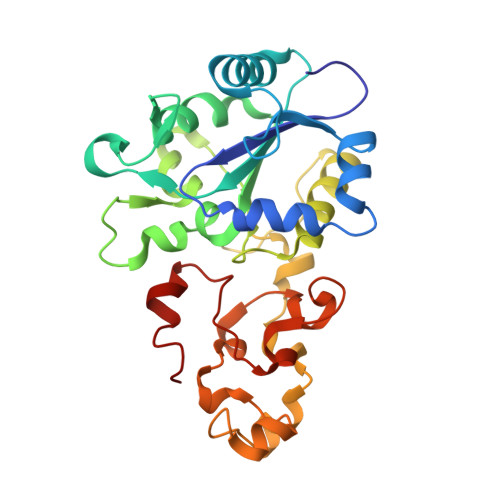
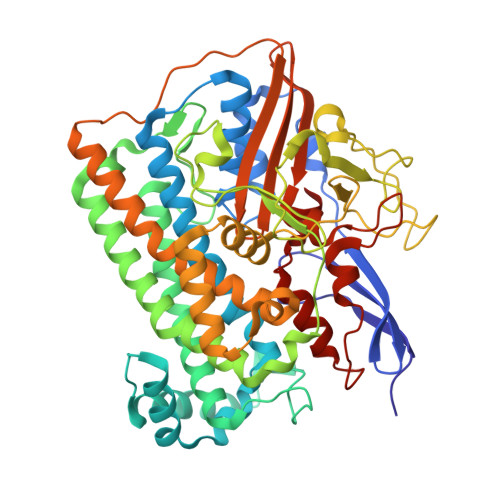
Removal of the bridging ligand atom at the Ni-Fe active site of [NiFe] hydrogenase upon reduction with H2, as revealed by X-ray structure analysis at 1.4 A resolution.
Higuchi, Y., Ogata, H., Miki, K., Yasuoka, N., Yagi, T.(1999) Structure 7: 549-556
- PubMed: 10378274
- DOI: https://doi.org/10.1016/s0969-2126(99)80071-9
- Primary Citation of Related Structures:
1H2R - PubMed Abstract:
The active site of [NiFe] hydrogenase, a heterodimeric protein, is suggested to be a binuclear Ni-Fe complex having three diatomic ligands to the Fe atom and three bridging ligands between the Fe and Ni atoms in the oxidized form of the enzyme. Two of the bridging ligands are thiolate sidechains of cysteinyl residues of the large subunit, but the third bridging ligand was assigned as a non-protein monatomic sulfur species in Desulfovibrio vulgaris Miyazaki F hydrogenase.
Organizational Affiliation:
Division of Chemistry, Graduate School of Science, Kyoto University, Japan. higuchi@kuchem.kyoto-u.ac.jp