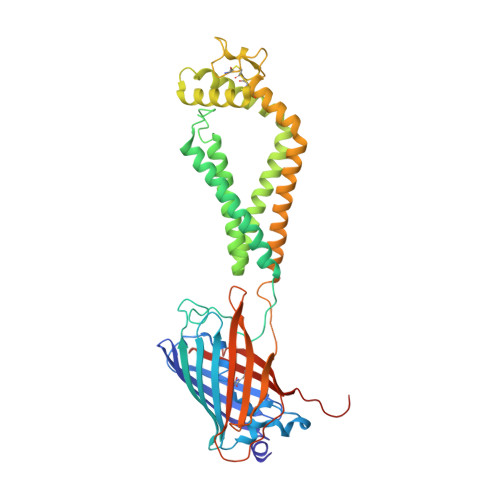
Open conformation of tetraspanins shapes interaction partner networks on cell membranes.
Yang, Y., Liu, X.R., Greenberg, Z.J., Zhou, F., He, P., Fan, L., Liu, S., Shen, G., Egawa, T., Gross, M.L., Schuettpelz, L.G., Li, W.(2020) EMBO J 39: e105246-e105246
- PubMed: 32974937
- DOI: https://doi.org/10.15252/embj.2020105246
- Primary Citation of Related Structures:
6WVG - PubMed Abstract:
Tetraspanins, including CD53 and CD81, regulate a multitude of cellular processes through organizing an interaction network on cell membranes. Here, we report the crystal structure of CD53 in an open conformation poised for partner interaction. The large extracellular domain (EC2) of CD53 protrudes away from the membrane surface and exposes a variable region, which is identified by hydrogen-deuterium exchange as the common interface for CD53 and CD81 to bind partners. The EC2 orientation in CD53 is supported by an extracellular loop (EC1). At the closed conformation of CD81, however, EC2 disengages from EC1 and rotates toward the membrane, thereby preventing partner interaction. Structural simulation shows that EC1-EC2 interaction also supports the open conformation of CD81. Disrupting this interaction in CD81 impairs the accurate glycosylation of its CD19 partner, the target for leukemia immunotherapies. Moreover, EC1 mutations in CD53 prevent the chemotaxis of pre-B cells toward a chemokine that supports B-cell trafficking and homing within the bone marrow, a major CD53 function identified here. Overall, an open conformation is required for tetraspanin-partner interactions to support myriad cellular processes.
- Department of Biochemistry and Molecular Biophysics, Washington University School of Medicine, St. Louis, MO, USA.
Organizational Affiliation: