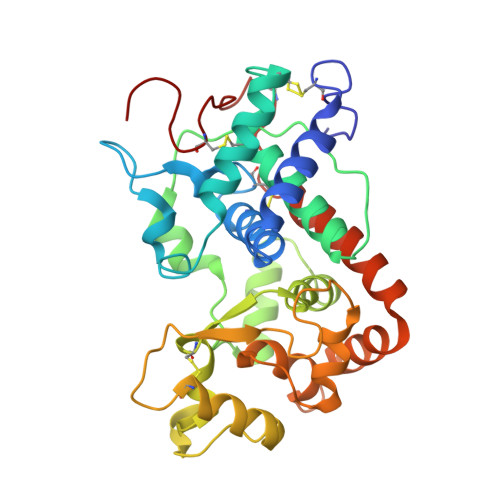
Post-Translational Modification and Extended Glycosylation Pattern of a Plant Latex Peroxidase of Native Source Characterized by X-Ray Crystallography.
Palm, G.J., Sharma, A., Kumari, M., Panjikar, S., Albrecht, D., Jagannadham, M.V., Hinrichs, W.(2014) FEBS J 281: 4319
- PubMed: 24980207
- DOI: https://doi.org/10.1111/febs.12900
- Primary Citation of Related Structures:
4CUO - PubMed Abstract:
The crystal structure of banyan peroxidase purified from the latex of Ficus benghalensis has been solved at 1.67 Å resolution by single-wavelength anomalous diffraction phasing. The refined structure includes 306 amino acid residues, a heme and two calcium ions. The protein belongs to class III peroxidases and is the first one from plant latex. Extensive glycosylation was observed with N-linked glycans attached to seven asparagine residues. The enzyme is stable with respect to a wide pH range, temperature, chemical denaturants and organic solvents, probably as a result of its high glycosylation. An unexpected post-translational modification of Asp290 was identified as succinimide moiety. Kinetic parameters of banyan peroxidase have been determined using various hydrogen donor substrates and hydrogen peroxide.
Organizational Affiliation:
Institut für Biochemie, Universität Greifswald, Germany.