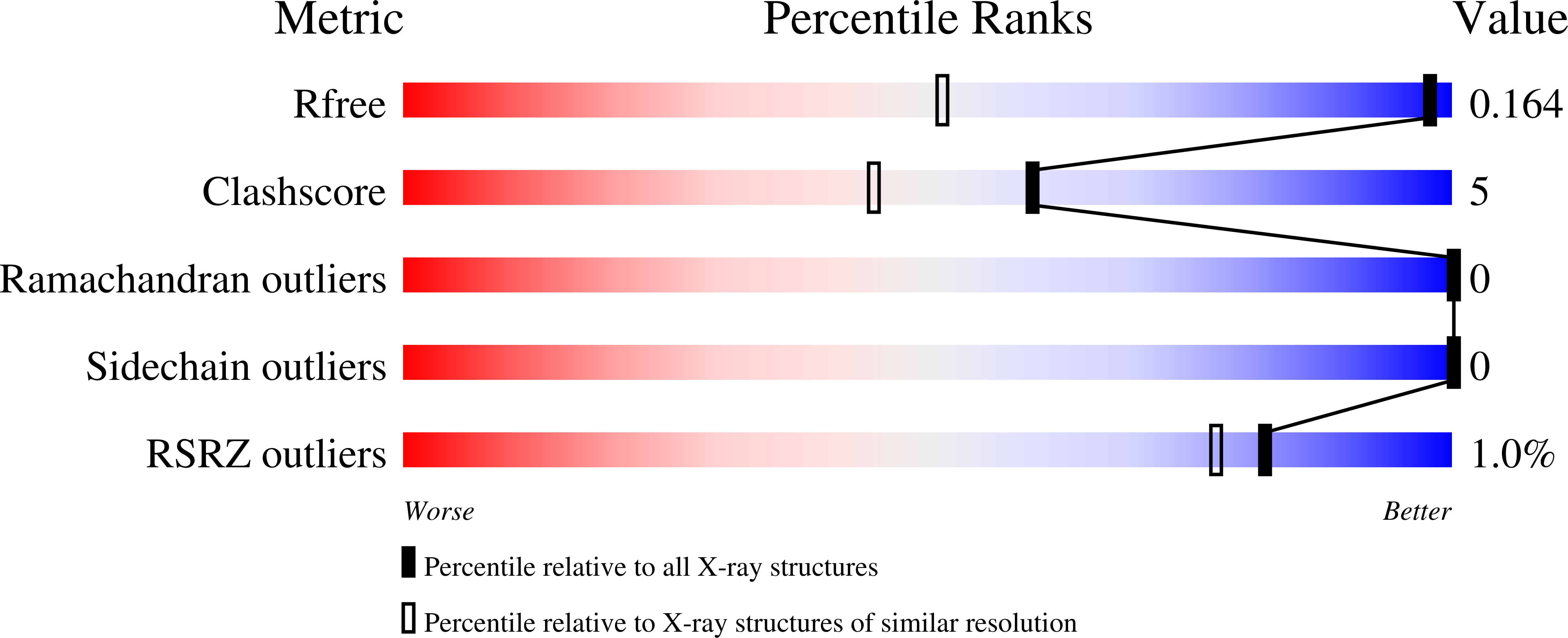
Enzymatic C-H activation of aromatic compounds through CO 2 fixation.
Aleku, G.A., Saaret, A., Bradshaw-Allen, R.T., Derrington, S.R., Titchiner, G.R., Gostimskaya, I., Gahloth, D., Parker, D.A., Hay, S., Leys, D.(2020) Nat Chem Biol 16: 1255-1260
- PubMed: 32719558
- DOI: https://doi.org/10.1038/s41589-020-0603-0
- Primary Citation of Related Structures:
6TIB, 6TIC, 6TIE, 6TIH, 6TIJ, 6TIL, 6TIN, 6TIO - PubMed Abstract:
The direct C-H carboxylation of aromatic compounds is an attractive route to the corresponding carboxylic acids, but remains challenging under mild conditions. It has been proposed that the first step in anaerobic microbial degradation of recalcitrant aromatic compounds is a UbiD-mediated carboxylation. In this study, we use the UbiD enzyme ferulic acid decarboxylase (Fdc) in combination with a carboxylic acid reductase to create aromatic degradation-inspired cascade reactions, leading to efficient functionalization of styrene through CO 2 fixation. We reveal that rational structure-guided laboratory evolution can expand the substrate scope of Fdc, resulting in activity on a range of mono- and bicyclic aromatic compounds through a single mutation. Selected variants demonstrated 150-fold improvement in the conversion of coumarillic acid to benzofuran + CO 2 and unlocked reactivity towards naphthoic acid. Our data demonstrate that UbiD-mediated C-H activation is a versatile tool for the transformation of aryl/alkene compounds and CO 2 into commodity chemicals.
Organizational Affiliation:
Manchester Institute of Biotechnology, Department of Chemistry, University of Manchester, Manchester, UK.