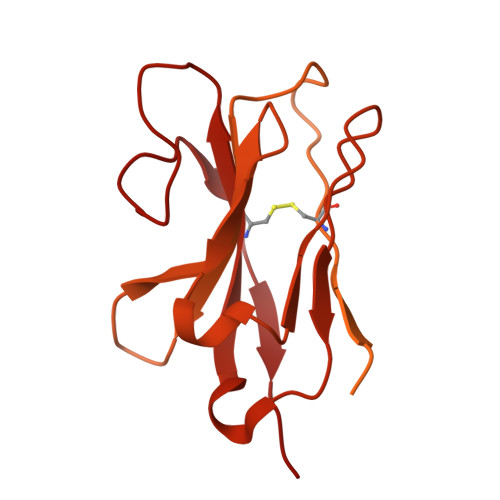
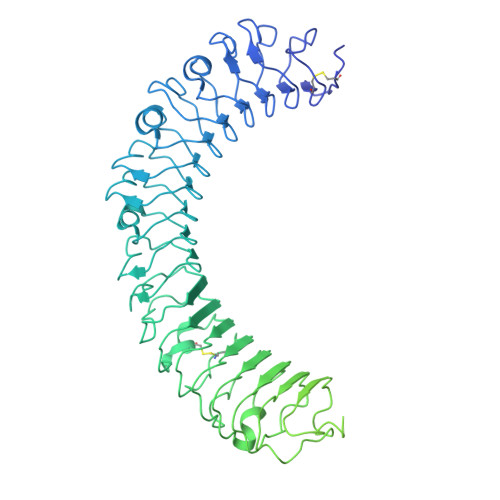
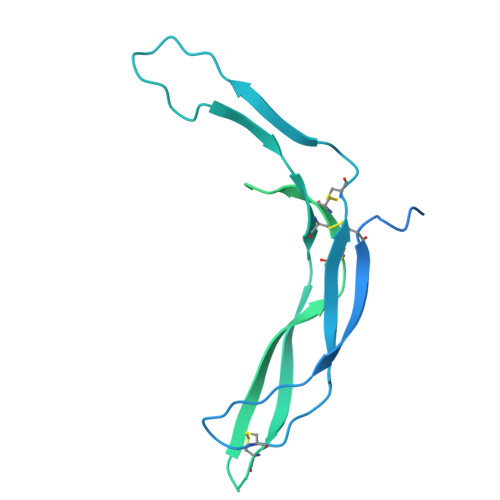
Distinct structural mechanisms of LGR4 modulation by Norrin and RSPOs in Wnt/ beta-catenin signaling.
Qiao, H., Hu, F., Wang, Y., Wang, L., Zhou, S., Guo, S., Xu, Y., Xu, J., Cui, Q., Yang, Q., Xu, H.E., Zhu, J., Geng, Y.(2025) Nat Commun 16: 6256-6256
- PubMed: 40624078 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-025-61545-z
- Primary Citation Related Structures:
9UOK - PubMed Abstract:
The Wnt/β-catenin pathway requires precise regulation for proper development and tissue homeostasis, yet the structural mechanisms enabling its fine-tuned control remain incompletely understood. Here, we reveal how LGR4 achieves differential signaling outcomes through distinct recognition of two key modulators: Norrin and R-spondins (RSPOs). Using cryo-electron microscopy, we determined the structure of full-length LGR4 bound to Norrin in a 2:2 stoichiometry, revealing a molecular bridging mechanism where Norrin dimer connect two LGR4 protomers in a spatial arrangement fundamentally distinct from the LGR4-RSPO2-ZNRF3 complex. Notably, Norrin binding to LGR4 sterically hinders simultaneous interaction with the Frizzled4 receptor, establishing a regulatory checkpoint in Wnt signaling. The partially overlapping binding sites for Norrin and RSPOs on LGR4 enable mutually exclusive interactions that drive distinct signaling outcomes. Disease-linked mutations map to distinct functional regions: those disrupting LGR4 interaction are associated with familial exudative vitreoretinopathy (FEVR), while others impairing Frizzled4 binding are linked to Norrie disease. Furthermore, we developed a high-affinity nanobody that blocks both Norrin and RSPO binding to LGR4, providing a potential tool for therapeutic intervention. These findings elucidate the structural basis of LGR4's dual signaling roles and lay the groundwork for therapeutic strategies targeting Wnt-related diseases.
- Engineering Research Center of Cell and Therapeutic Antibody, Ministry of Education, School of Pharmacy, Shanghai Jiao Tong University, Shanghai, China.
Organizational Affiliation: