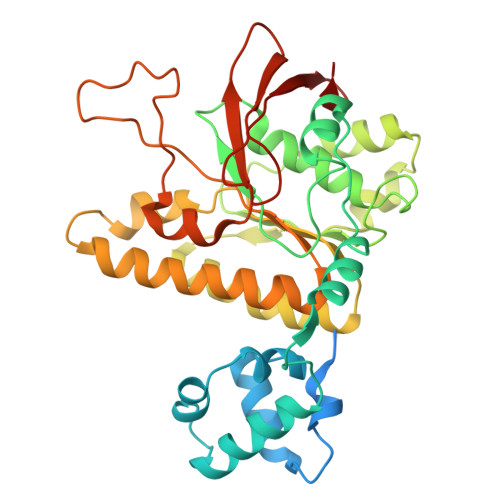
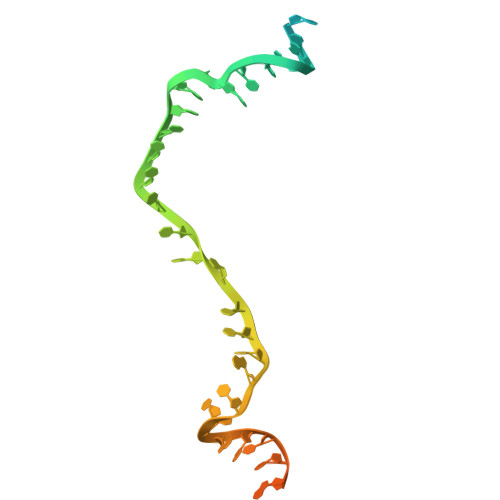
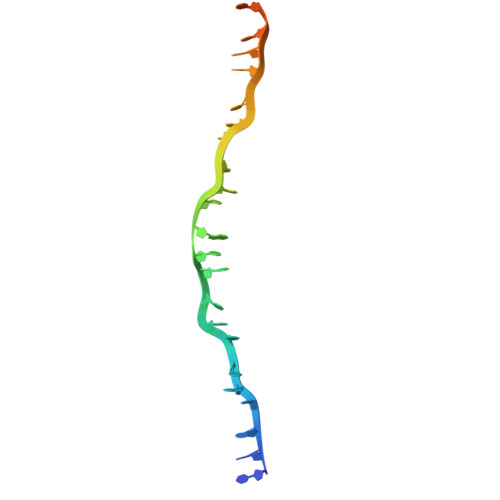
RAD51 D-loop structures reveal the mechanism of eukaryotic RAD51-mediated strand exchange.
Luo, S.C., Yang, C.H., Yeh, H.Y., Lin, C.W., Yeh, M.C., Chi, P., Ho, M.C.(2025) Nat Commun 17: 243-243
- PubMed: 41326376 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-025-66925-z
- Primary Citation Related Structures:
9UI4, 9UI5, 9UI7, 9UI8, 9UI9 - PubMed Abstract:
Strand exchange is a key step in homologous recombination, enabling template-based repair of DNA double-strand breaks. Eukaryotic RAD51 forms an ATP-dependent helical presynaptic filament on single-stranded DNA (ssDNA), which then searches for homologous double-stranded DNA (dsDNA), and catalyzes the strand exchange to form a D-loop in an ATP hydrolysis-independent manner. The molecular mechanism by which RAD51 facilitates dsDNA unwinding and pairing remains unclear. Here, we present cryo-EM structures of RAD51 mini-filaments bound to homologous dsDNA, capturing five intermediates from dsDNA recruitment to D-loop formation. These structures, together with molecular dynamics simulations, suggest a stepwise mechanism: the conserved N-terminal domain (NTD) recruits and bends the dsDNA, weakening base pairing near the exchange site. Subsequent engagement with positively-charged regions, including the loop L2 and loop Arg303-Arg306, further bends the homologous dsDNA, thereby not only positioning it closer to the strand exchange site but also inducing local base-pair opening. Additionally, the loop L2 (Met278 and Phe279) inserts between strands, and the secondary DNA binding sites (S2 sites) capture the displaced strand to prevent strand reannealing. Together, our findings provide detailed insight into a spatially coordinated mechanism of strand exchange by RAD51.
- Institute of Biological Chemistry, Academia Sinica, Taipei, Taiwan.
Organizational Affiliation: