Structural remodeling of the mitochondrial protein biogenesis machinery upon proteostatic stress
Ehses, K., Lopez-Alonso, J.P., Gonzalez, A., Tascon, I., Ubarretxena-Belandia, I., Fernandez-Busnadiego, R.To be published.
Experimental Data Snapshot
Starting Models: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 60 kDa heat shock protein, mitochondrial | 527 | Homo sapiens | Mutation(s): 0 Gene Names: HSPD1, HSP60 EC: 5.6.1.7 | 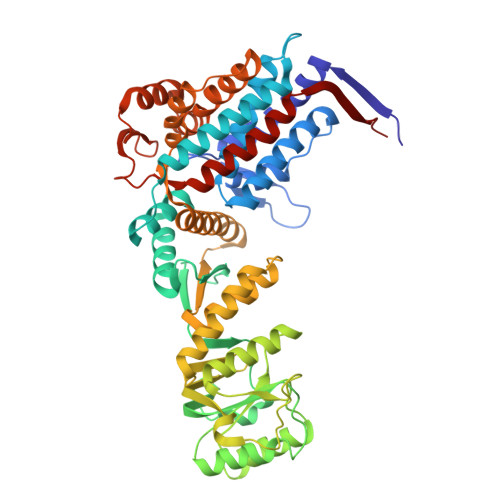 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P10809 GTEx: ENSG00000144381 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P10809 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 10 kDa heat shock protein, mitochondrial | 102 | Homo sapiens | Mutation(s): 0 Gene Names: HSPE1 | 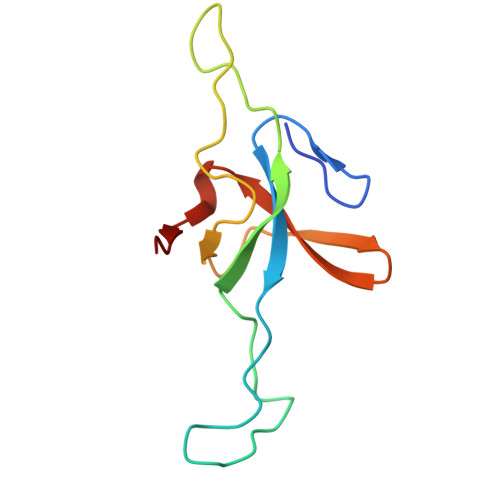 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P61604 GTEx: ENSG00000115541 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P61604 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ATP (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | BA [auth C] CB [auth L] EA [auth D] FB [auth M] HA [auth E] | ADENOSINE-5'-TRIPHOSPHATE C10 H16 N5 O13 P3 ZKHQWZAMYRWXGA-KQYNXXCUSA-N |  | ||
| K (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | AA [auth B] BB [auth K] DA [auth C] EB [auth L] GA [auth D] | POTASSIUM ION K NPYPAHLBTDXSSS-UHFFFAOYSA-N |  | ||
| MG (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | AB [auth K] CA [auth C] DB [auth L] FA [auth D] GB [auth M] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Task | Software Package | Version |
|---|---|---|
| MODEL REFINEMENT | PHENIX | 1.21rc1_4933 |
| RECONSTRUCTION | cryoSPARC | 4.1.2 |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Ministerio de Ciencia e Innovacion (MCIN) | Spain | PID2022-143177NB-I00 |