Engineered coagulation factor VIII with enhanced secretion and coagulation potential for hemophilia A gene therapy.
Kashiwakura, Y., Nakajima, Y., Horinaka, K., Lopes, T.J.S., Furuta, Y., Yamaguchi, Y., Baatartsogt, N., Hayakawa, M., Katakai, Y., Uchiyama, S., Nureki, O., Nogami, K., Ohmori, T.(2026) Blood 147: 402-415
- PubMed: 41198051 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1182/blood.2025028481
- Primary Citation Related Structures:
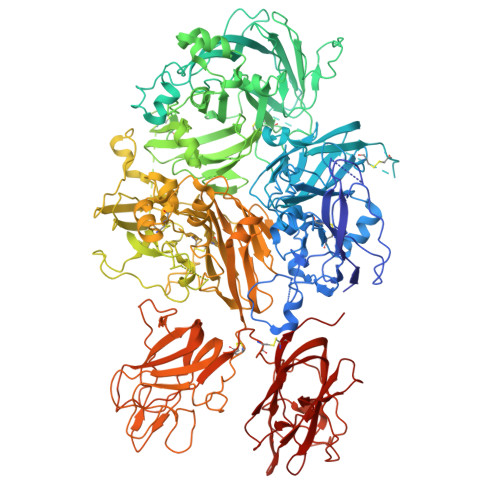
9JBV - PubMed Abstract:
The major challenges of gene therapy for hemophilia A using adeno-associated virus (AAV) vectors are reducing vector doses and the long-term maintenance of stable factor VIII (FVIII). In this study, we developed engineered human B-domain-deleted FVIIIs (FVIIISQ) with enhanced secretion and coagulation potential. Intracellular accumulation was markedly reduced in some engineered FVIIISQ, resulting in reduced unfolded protein responses. The administration of AAV vectors carrying engineered FVIIISQ to hemophilia A mice resulted in ∼8-fold higher FVIII activity and 4-fold higher FVIII antigen levels compared with wild-type FVIIISQ administration. The specific FVIII activity of the engineered FVIIISQ was 3.6 times higher than that of the wild-type FVIIISQ, and its binding to activated coagulation factor IX was significantly enhanced, which is supported by the structural analysis. In macaques, the administration of AAV5 vector carrying the engineered FVIIISQ without CpG sequences resulted in a supraphysiological increase in plasma FVIII activity at a dose one-thirtieth that of valoctocogene roxaparvovec (2 × 1012 vector genome per kg). The engineered FVIIISQ may thus provide stable, long-term therapeutic efficacy in AAV-mediated hemophilia A gene therapy even at low doses.
- Department of Biochemistry, Jichi Medical University School of Medicine, Shimotsuke, Japan.
Organizational Affiliation: