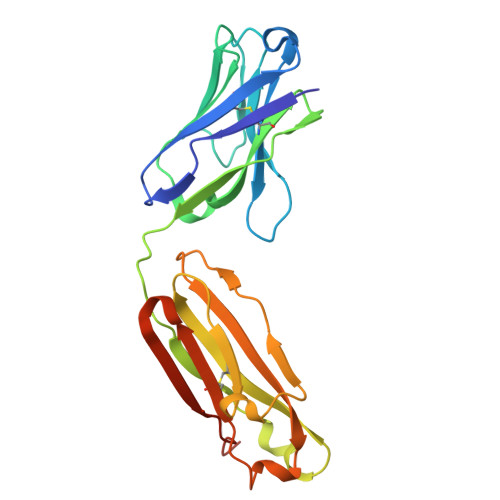
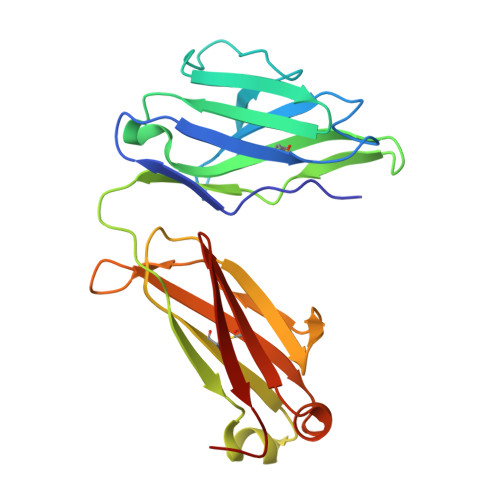
In vivo efficacy of human anti-preS2 HBV neutralizing antibodies
Beretta, M., Haouz, A., Mechaly, A.E., Caillet-Saguy, C., Planchais, C., Szerman, N., Aronthippaitoon, Y., Ungeheuer, M.-N., Pol, S., Gaudy-graffin, C., Sureau, C., Bourgine, M., Mouquet, H.To be published.