Cryo-EM structure of the SbmA in the inward-facing closed conformation bound to 2 sybodies
Thangaratnarajah, C., Ettema, T.W., Slotboom, D.J.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Peptide antibiotic transporter SbmA | 426 | Escherichia coli | Mutation(s): 0 Gene Names: sbmA, b0377, JW0368 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0AFY6 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Sybody 2 | 146 | synthetic construct | Mutation(s): 0 | 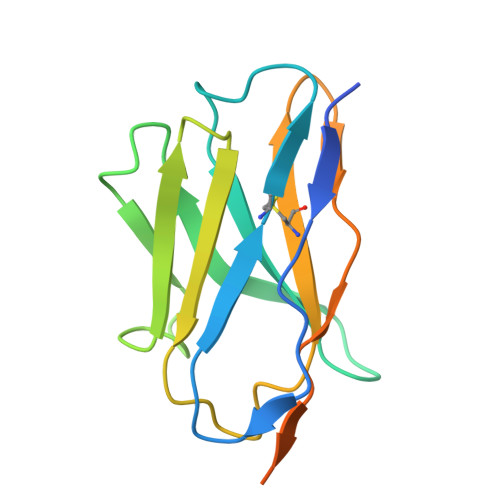 | |
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| PGT (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | E [auth A], F [auth B] | (1S)-2-{[{[(2R)-2,3-DIHYDROXYPROPYL]OXY}(HYDROXY)PHOSPHORYL]OXY}-1-[(PALMITOYLOXY)METHYL]ETHYL STEARATE C40 H79 O10 P KBPVYRBBONZJHF-AMAPPZPBSA-N |  | ||
| Task | Software Package | Version |
|---|---|---|
| RECONSTRUCTION | cryoSPARC | 3.3.2 |
| MODEL REFINEMENT | PHENIX | 1.20.1-4487 |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Netherlands Organisation for Scientific Research (NWO) | Netherlands | 714.018.003 |
| Netherlands Organisation for Scientific Research (NWO) | Netherlands | 024.003.019 |
| Netherlands Organisation for Scientific Research (NWO) | Netherlands | 184.034.014 |
| European Union (EU) | European Union | 847675 |