Mechanistic insights into the structure-based design of a CspZ-targeting Lyme disease vaccine.
Brangulis, K., Malfetano, J., Marcinkiewicz, A.L., Wang, A., Chen, Y.L., Lee, J., Liu, Z., Yang, X., Strych, U., Tupina, D., Akopjana, I., Bottazzi, M.E., Pal, U., Hsieh, C.L., Chen, W.H., Lin, Y.P.(2025) Nat Commun 16: 2898-2898
- PubMed: 40189575 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-025-58182-x
- Primary Citation Related Structures:
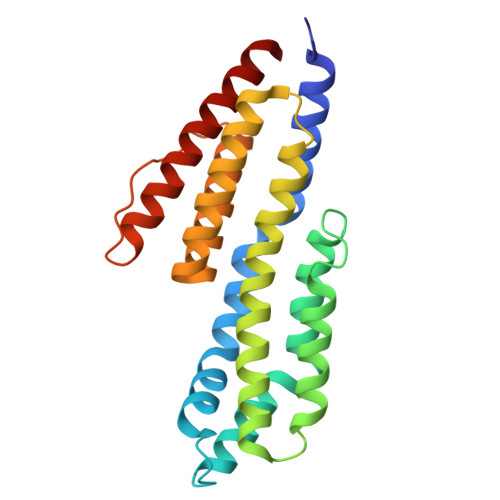
9F1V, 9F21 - PubMed Abstract:
Borrelia burgdorferi (Bb) causes Lyme disease (LD), one of the most common vector-borne diseases in the Northern Hemisphere. Here, we solve the crystal structure of a mutated Bb vaccine antigen, CspZ-YA that lacks the ability to bind to host complement factor H (FH). We generate point mutants of CspZ-YA and identify CspZ-YA I183Y and CspZ-YA C187S to trigger more robust bactericidal responses. Compared to CspZ-YA, these CspZ-YA mutants require a lower immunization frequency to protect mice from LD-associated inflammation and bacterial colonization. Antigenicity of wild-type and mutant CspZ-YA proteins are similar, as measured using sera from infected people or immunized female mice. Structural comparison of CspZ-YA with CspZ-YA I183Y and CspZ-YA C187S shows enhanced interactions of two helices adjacent to the FH-binding sites in the mutants, consistent with their elevated thermostability. In line with these findings, protective CspZ-YA monoclonal antibodies show increased binding to CspZ-YA at a physiological temperature (37 °C). In summary, this proof-of-concept study applies structural vaccinology to enhance intramolecular interactions for the long-term stability of a Bb antigen while maintaining its protective epitopes, thus promoting LD vaccine development.
- Latvian Biomedical Research and Study Centre, Riga, Latvia. kalvis@biomed.lu.lv.
Organizational Affiliation: