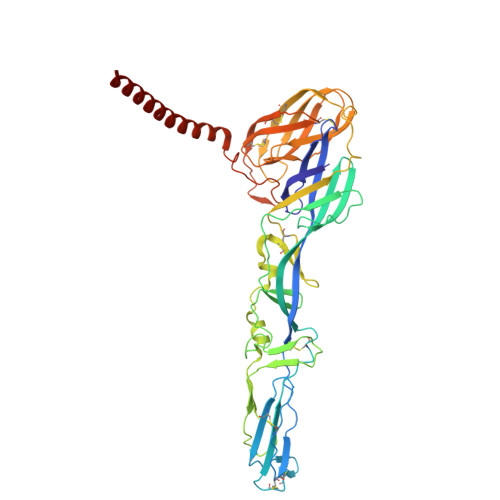
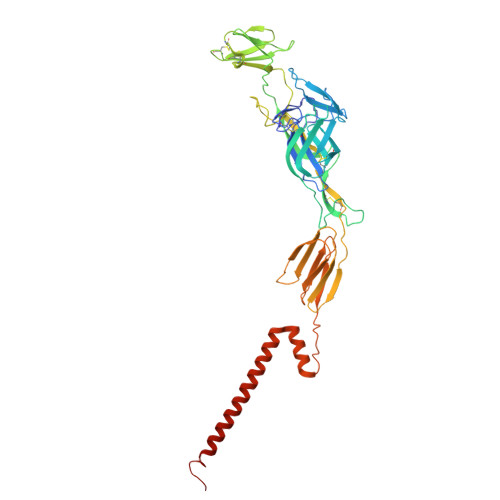
Molecular basis for shifted receptor recognition by an encephalitic arbovirus.
Fan, X., Li, W., Oros, J., Plante, J.A., Mitchell, B.M., Plung, J.S., Basu, H., Nagappan-Chettiar, S., Boeckers, J.M., Tjang, L.V., Mann, C.J., Brusic, V., Buck, T.K., Varnum, H., Yang, P., Malcolm, L.M., Choi, S.Y., de Souza, W.M., Chiu, I.M., Umemori, H., Weaver, S.C., Plante, K.S., Abraham, J.(2025) Cell 188: 2957-2973.e28
- PubMed: 40187345 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.cell.2025.03.029
- Primary Citation Related Structures:
9DQV, 9DQX, 9DQY, 9DQZ - PubMed Abstract:
Western equine encephalitis virus (WEEV) is an arbovirus that historically caused large outbreaks of encephalitis throughout the Americas. WEEV binds protocadherin 10 (PCDH10) as a receptor, and highly virulent ancestral WEEV strains also bind low-density lipoprotein receptor (LDLR)-related proteins. As WEEV declined as a human pathogen in North America over the past century, isolates have lost the ability to bind mammalian receptors while still recognizing avian receptors. To explain shifts in receptor dependencies and assess the risk of WEEV re-emergence, we determined cryoelectron microscopy structures of WEEV bound to human PCDH10, avian PCDH10, and human very-low-density lipoprotein receptor (VLDLR). We show that one to three E2 glycoprotein substitutions are sufficient for a nonpathogenic strain to regain the ability to bind mammalian receptors. A soluble VLDLR fragment protects mice from lethal challenge by a virulent ancestral WEEV strain. Because WEEV recently re-emerged in South America after decades of inactivity, our findings have important implications for outbreak preparedness.
- Department of Microbiology, Blavatnik Institute, Harvard Medical School, Boston, MA, USA.
Organizational Affiliation: