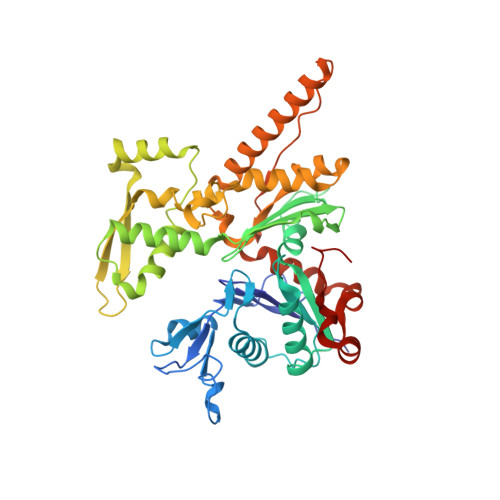
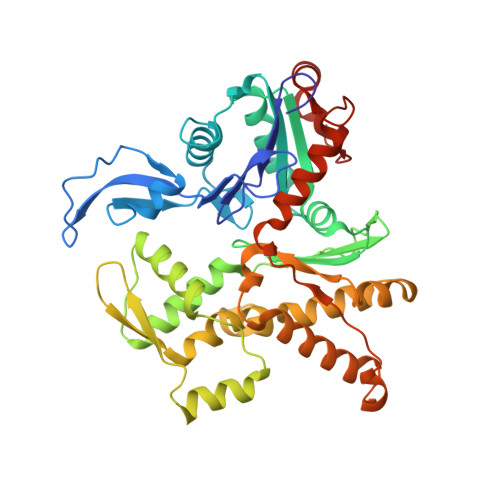
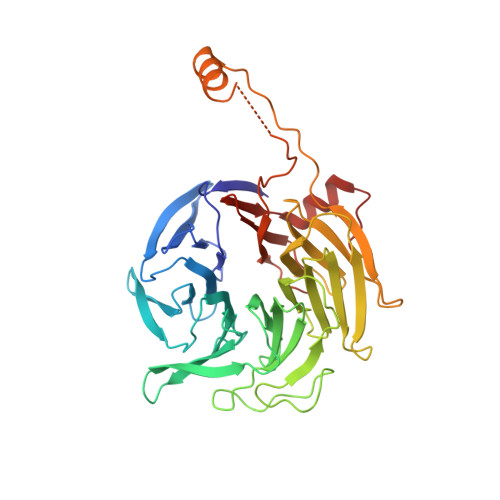
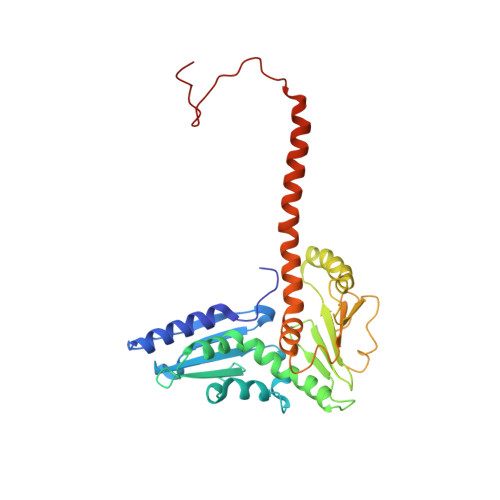
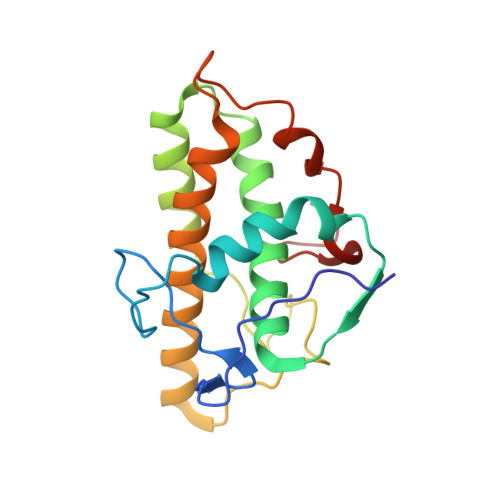
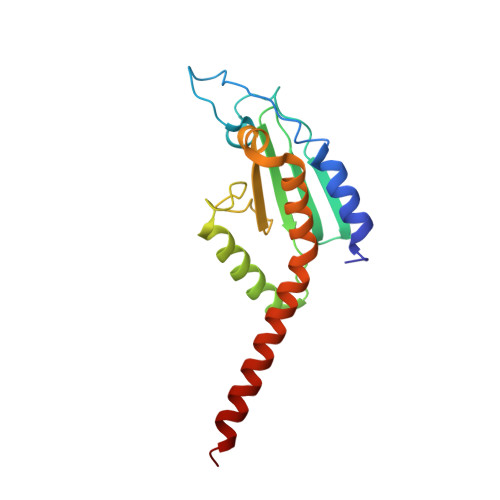
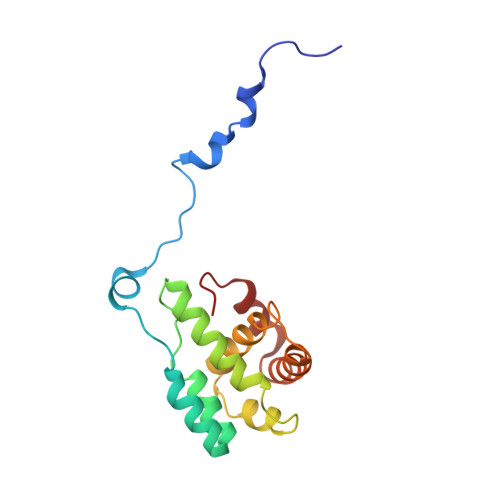
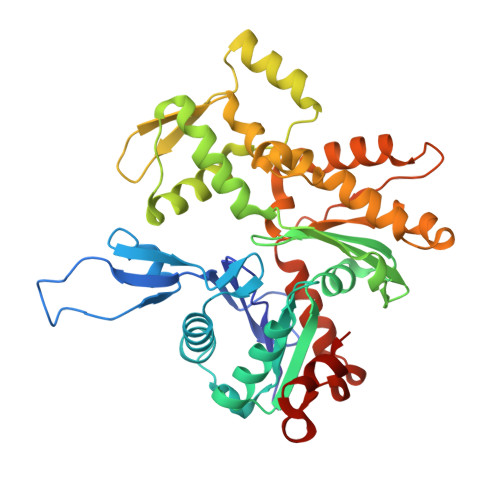
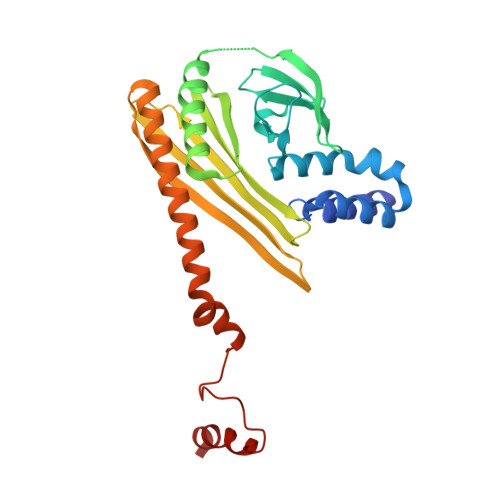
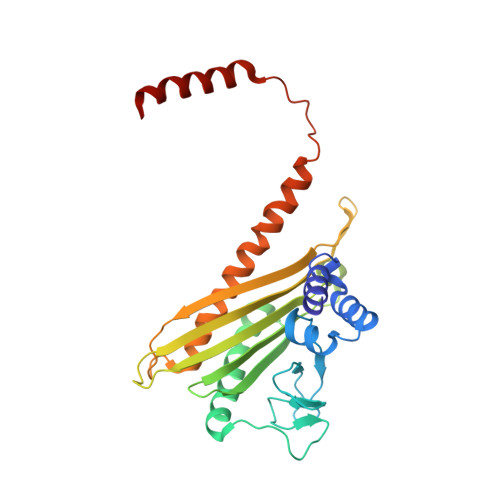
Cortactin stabilizes actin branches by bridging activated Arp2/3 to its nucleated actin filament.
Liu, T., Cao, L., Mladenov, M., Jegou, A., Way, M., Moores, C.A.(2024) Nat Struct Mol Biol 31: 801-809
- PubMed: 38267598 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41594-023-01205-2
- Primary Citation Related Structures:
8P94 - PubMed Abstract:
Regulation of the assembly and turnover of branched actin filament networks nucleated by the Arp2/3 complex is essential during many cellular processes, including cell migration and membrane trafficking. Cortactin is important for actin branch stabilization, but the mechanism by which this occurs is unclear. Given this, we determined the structure of vertebrate cortactin-stabilized Arp2/3 actin branches using cryogenic electron microscopy. We find that cortactin interacts with the new daughter filament nucleated by the Arp2/3 complex at the branch site, rather than the initial mother actin filament. Cortactin preferentially binds activated Arp3. It also stabilizes the F-actin-like interface of activated Arp3 with the first actin subunit of the new filament, and its central repeats extend along successive daughter-filament subunits. The preference of cortactin for activated Arp3 explains its retention at the actin branch and accounts for its synergy with other nucleation-promoting factors in regulating branched actin network dynamics.
- Institute of Structural and Molecular Biology, Birkbeck College, London, UK.
Organizational Affiliation: