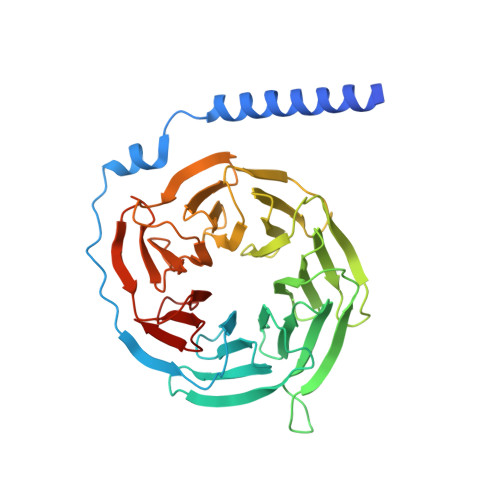
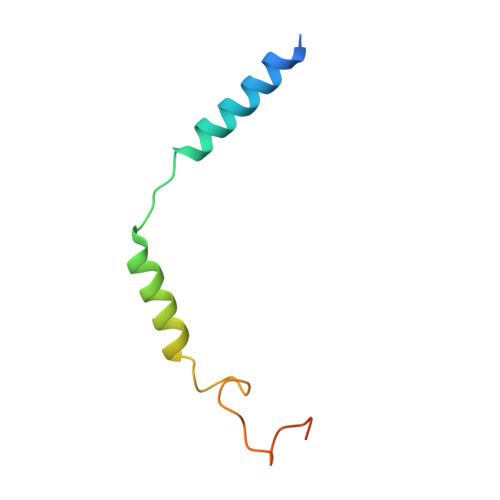
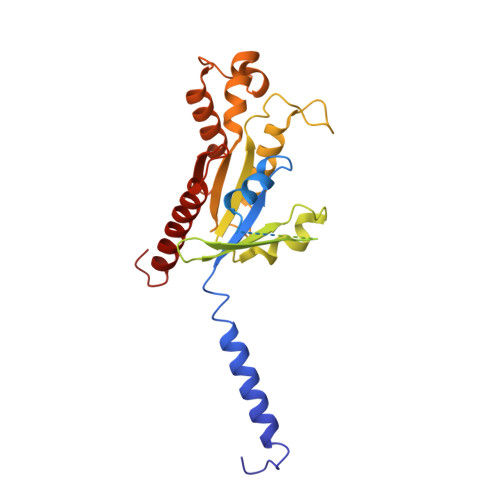
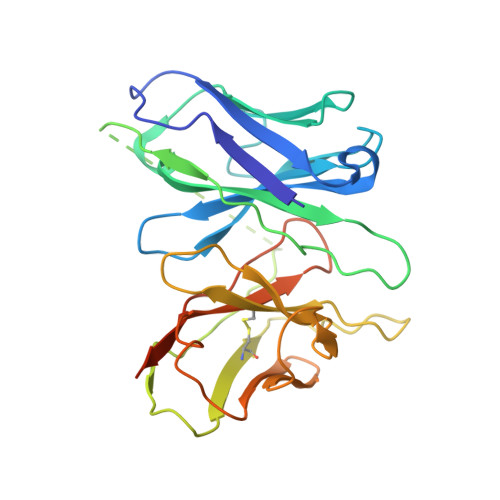
Biased allosteric activation of ketone body receptor HCAR2 suppresses inflammation.
Zhao, C., Wang, H., Liu, Y., Cheng, L., Wang, B., Tian, X., Fu, H., Wu, C., Li, Z., Shen, C., Yu, J., Yang, S., Hu, H., Fu, P., Ma, L., Wang, C., Yan, W., Shao, Z.(2023) Mol Cell 83: 3171
- PubMed: 37597514 Search on PubMed
- DOI: https://doi.org/10.1016/j.molcel.2023.07.030
- Primary Citation Related Structures:
8JHY, 8JII, 8JIL, 8JIM - PubMed Abstract:
Hydroxycarboxylic acid receptor 2 (HCAR2), modulated by endogenous ketone body β-hydroxybutyrate and exogenous niacin, is a promising therapeutic target for inflammation-related diseases. HCAR2 mediates distinct pathophysiological events by activating G i/o protein or β-arrestin effectors. Here, we characterize compound 9n as a G i -biased allosteric modulator (BAM) of HCAR2 and exhibit anti-inflammatory efficacy in RAW264.7 macrophages via a specific HCAR2-G i pathway. Furthermore, four structures of HCAR2-G i complex bound to orthosteric agonists (niacin or monomethyl fumarate), compound 9n, and niacin together with compound 9n simultaneously reveal a common orthosteric site and a unique allosteric site. Combined with functional studies, we decipher the action framework of biased allosteric modulation of compound 9n on the orthosteric site. Moreover, co-administration of compound 9n with orthosteric agonists could enhance anti-inflammatory effects in the mouse model of colitis. Together, our study provides insight to understand the molecular pharmacology of the BAM and facilitates exploring the therapeutic potential of the BAM with orthosteric drugs.
- Division of Nephrology and Kidney Research Institute, State Key Laboratory of Biotherapy and Cancer Center, West China Hospital, Sichuan University, Chengdu 610041, Sichuan, China; Frontiers Medical Center, Tianfu Jincheng Laboratory, Chengdu 610212, Sichuan, China.
Organizational Affiliation: