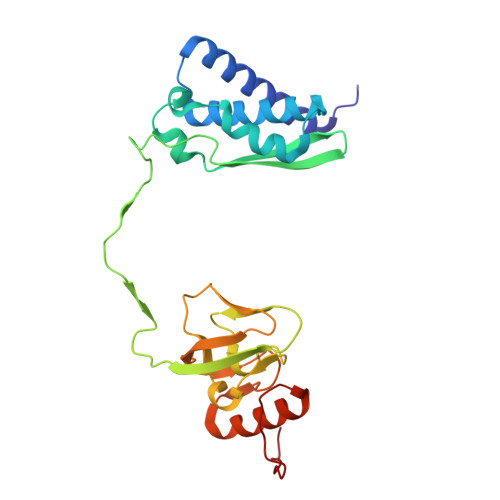
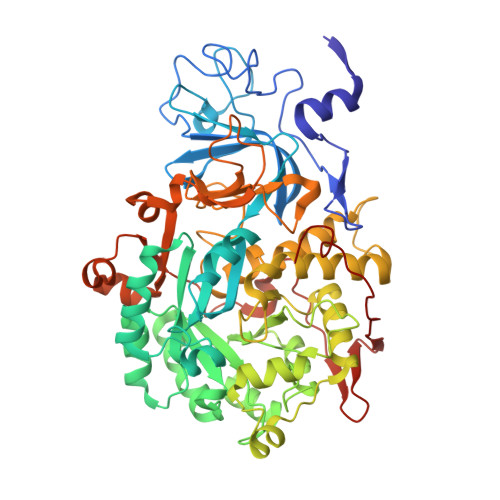
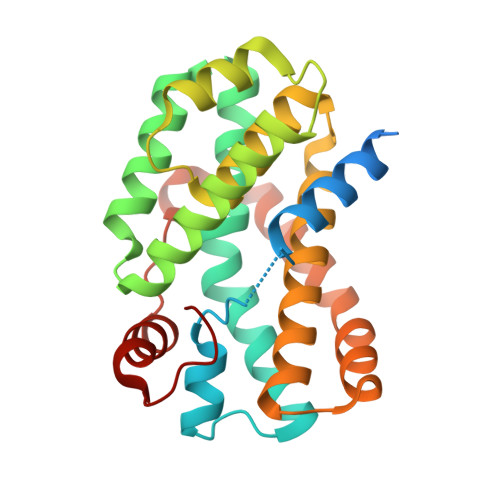
Delivering a toxic metal to the active site of urease.
Nim, Y.S., Fong, I.Y.H., Deme, J., Tsang, K.L., Caesar, J., Johnson, S., Pang, L.T.H., Yuen, N.M.H., Ng, T.L.C., Choi, T., Wong, Y.Y.H., Lea, S.M., Wong, K.B.(2023) Sci Adv 9: eadf7790-eadf7790
- PubMed: 37083535
- DOI: https://doi.org/10.1126/sciadv.adf7790
- Primary Citation Related Structures:
8HC1, 8HCN - PubMed Abstract:
Urease is a nickel (Ni) enzyme that is essential for the colonization of Helicobacter pylori in the human stomach. To solve the problem of delivering the toxic Ni ion to the active site without diffusing into the cytoplasm, cells have evolved metal carrier proteins, or metallochaperones, to deliver the toxic ions to specific protein complexes. Ni delivery requires urease to form an activation complex with the urease accessory proteins UreFD and UreG. Here, we determined the cryo-electron microscopy structures of H. pylori UreFD/urease and Klebsiella pneumoniae UreD/urease complexes at 2.3- and 2.7-angstrom resolutions, respectively. Combining structural, mutagenesis, and biochemical studies, we show that the formation of the activation complex opens a 100-angstrom-long tunnel, where the Ni ion is delivered through UreFD to the active site of urease.
- School of Life Sciences, Centre for Protein Science and Crystallography, State Key Laboratory of Agrobiotechnology, The Chinese University of Hong Kong, Hong Kong, China.
Organizational Affiliation: