Crystal structure of actinomycin D and Echinomycin-d(ACGGGCT/AGCCCCGT) complex
Lee, Y.Y., Hou, M.H.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Echinomycin | E, H [auth A] | 8 | Streptomyces | Mutation(s): 0 | 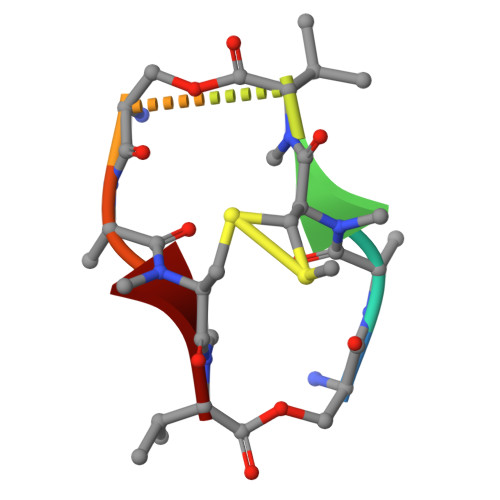 |
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| actinomycin D | F [auth I], G [auth D] | 11 | Streptomyces | Mutation(s): 0 | 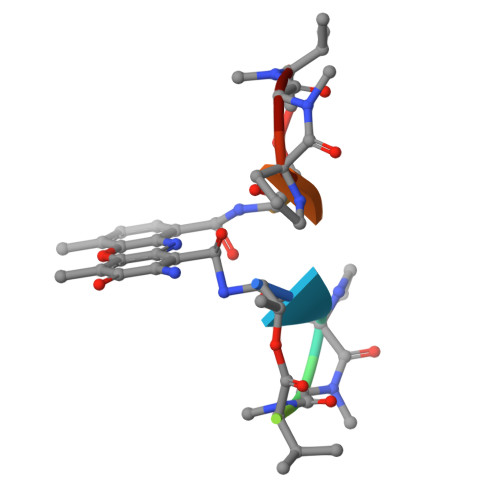 |
Entity ID: 1 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| DNA (5'-D(P*AP*GP*CP*CP*CP*CP*GP*T)-3') | A [auth B], C [auth K] | 8 | synthetic construct |  |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
Entity ID: 2 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| DNA (5'-D(P*AP*CP*GP*GP*GP*CP*T)-3') | B [auth C], D [auth L] | 7 | synthetic construct |  |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| QUI (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | I [auth E], J [auth E], K [auth A], L [auth A] | 2-CARBOXYQUINOXALINE C9 H6 N2 O2 UPUZGXILYFKSGE-UHFFFAOYSA-N |  | ||
| Modified Residues 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| DSN Query on DSN | E, H [auth A] | D-PEPTIDE LINKING | C3 H7 N O3 |  | -- |
| MVA Query on MVA | E, H [auth A] | L-PEPTIDE LINKING | C6 H13 N O2 |  | VAL |
| N2C Query on N2C | E, H [auth A] | L-PEPTIDE LINKING | C5 H11 N O2 S |  | -- |
| NCY Query on NCY | E, H [auth A] | L-PEPTIDE LINKING | C4 H9 N O2 S |  | -- |
| DVA Query on DVA | F [auth I], G [auth D] | D-PEPTIDE LINKING | C5 H11 N O2 |  | -- |
| SAR Query on SAR | F [auth I], G [auth D] | PEPTIDE LINKING | C3 H7 N O2 |  | GLY |
| Entity ID: 3 | |||||
|---|---|---|---|---|---|
| ID | Chains | Name | Type/Class | 2D Diagram | 3D Interactions |
| PRD_000491 Query on PRD_000491 | E [auth A], H [auth E] | Echinomycin | Cyclic depsipeptide / Antibiotic |  | |
| Entity ID: 4 | |||||
|---|---|---|---|---|---|
| ID | Chains | Name | Type/Class | 2D Diagram | 3D Interactions |
| PRD_000001 Query on PRD_000001 | F [auth D], G [auth I] | Actinomycin D | Polypeptide / Antibiotic |  | |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 132.661 | α = 90 |
| b = 132.661 | β = 90 |
| c = 50.052 | γ = 120 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-2000 | data scaling |
| HKL-2000 | data reduction |
| PHENIX | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Ministry of Science and Technology (MoST, Taiwan) | Taiwan | -- |