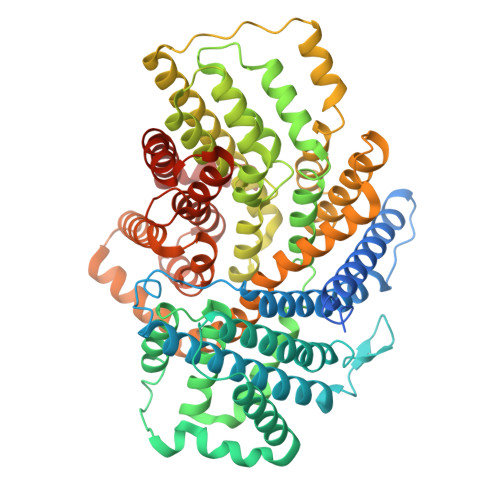
Molecular determinants of Neu5Ac binding to a tripartite ATP independent periplasmic (TRAP) transporter.
Goyal, P., Dhanabalan, K., Scalise, M., Friemann, R., Indiveri, C., Dobson, R.C.J., Vinothkumar, K.R., Ramaswamy, S.(2025) Elife 13
- PubMed: 39912804 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.7554/eLife.98158
- Primary Citation Related Structures:
8Y4W, 8Y4X - PubMed Abstract:
N -Acetylneuraminic acid (Neu5Ac) is a negatively charged nine-carbon amino sugar that is often the peripheral sugar in human cell-surface glycoconjugates. Some bacteria scavenge, import, and metabolize Neu5Ac or redeploy it on their cell surfaces for immune evasion. The import of Neu5Ac by many bacteria is mediated by tripartite ATP-independent periplasmic (TRAP) transporters. We have previously reported the structures of SiaQM, a membrane-embedded component of the Haemophilus influenzae TRAP transport system, (Currie et al., 2024). However, none of the published structures contain Neu5Ac bound to SiaQM. This information is critical for defining the transport mechanism and for further structure-activity relationship studies. Here, we report the structures of Fusobacterium nucleatum SiaQM with and without Neu5Ac. Both structures are in an inward (cytoplasmic side) facing conformation. The Neu5Ac-bound structure reveals the interactions of Neu5Ac with the transporter and its relationship with the Na + binding sites. Two of the Na + -binding sites are similar to those described previously. We identify a third metal-binding site that is further away and buried in the elevator domain. Ser300 and Ser345 interact with the C1-carboxylate group of Neu5Ac. Proteoliposome-based transport assays showed that Ser300-Neu5Ac interaction is critical for transport, whereas Ser345 is dispensable. Neu5Ac primarily interacts with residues in the elevator domain of the protein, thereby supporting the elevator with an operator mechanism. The residues interacting with Neu5Ac are conserved, providing fundamental information required to design inhibitors against this class of proteins.
- Biochemical Sciences Division, CSIR-National Chemical Laboratory, Pune, India.
Organizational Affiliation: