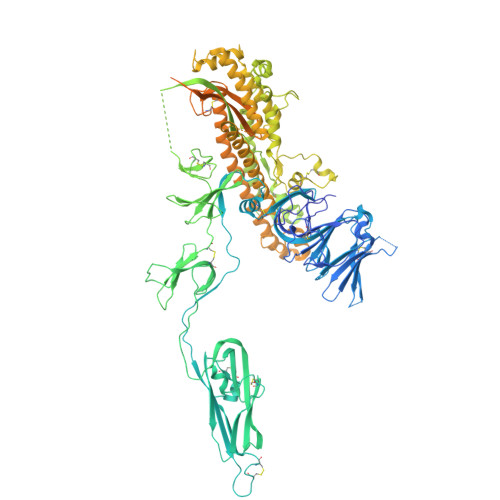
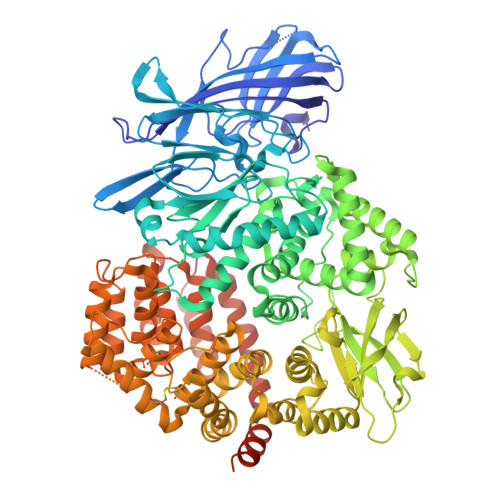
Molecular basis of host recognition of human coronavirus 229E.
Tsai, Y.X., Chien, Y.C., Hsu, M.F., Khoo, K.H., Hsu, S.D.(2025) Nat Commun 16: 2045-2045
- PubMed: 40016196 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-025-57359-8
- Primary Citation Related Structures:
8WDE - PubMed Abstract:
Human coronavirus 229E (HCoV-229E) is the earliest CoV found to infect humans. It binds to the human aminopeptidase N (hAPN) through the receptor binding domain (RBD) of its spike (S) protein to achieve host recognition. We present the cryo-electron microscopy structure of two HCoV-229E S protein in complex with a dimeric hAPN to provide structural insights on how the HCoV-229E S protein opens up its RBD to engage with its host receptor, information that is currently missing among alphacoronaviruses to which HCoV-229E belong. We quantitatively profile the glycosylation of HCoV-229E S protein and hAPN to deduce the glyco-shielding effects pertinent to antigenicity and host recognition. Finally, we present an atomic model of fully glycosylated HCoV-229E S in complex with hAPN anchored on their respective membrane bilayers to recapitulate the structural basis of the first step of host infection by HCoV-229E.
- Institute of Biological Chemistry, Academia Sinica, Taipei, 11529, Taiwan.
Organizational Affiliation: