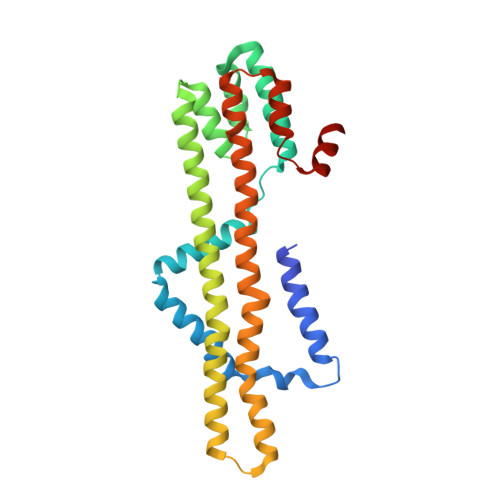
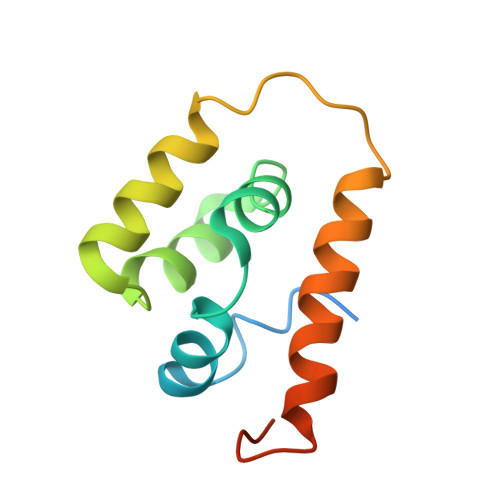
Structural basis of directional switching by the bacterial flagellum.
Johnson, S., Deme, J.C., Furlong, E.J., Caesar, J.J.E., Chevance, F.F.V., Hughes, K.T., Lea, S.M.(2024) Nat Microbiol 9: 1282-1292
- PubMed: 38459206 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41564-024-01630-z
- Primary Citation Related Structures:
8UCS, 8UMD, 8UMX, 8UOX, 8UPL - PubMed Abstract:
The bacterial flagellum is a macromolecular protein complex that harvests energy from uni-directional ion flow across the inner membrane to power bacterial swimming via rotation of the flagellar filament. Rotation is bi-directional, with binding of a cytoplasmic chemotactic response regulator controlling reversal, though the structural and mechanistic bases for rotational switching are not well understood. Here we present cryoelectron microscopy structures of intact Salmonella flagellar basal bodies (3.2-5.5 Å), including the cytoplasmic C-ring complexes required for power transmission, in both counter-clockwise and clockwise rotational conformations. These reveal 180° movements of both the N- and C-terminal domains of the FliG protein, which, when combined with a high-resolution cryoelectron microscopy structure of the MotA 5 B 2 stator, show that the stator shifts from the outside to the inside of the C-ring. This enables rotational switching and reveals how uni-directional ion flow across the inner membrane is used to accomplish bi-directional rotation of the flagellum.
- Center for Structural Biology, CCR, NCI, Frederick, MD, USA. steven.johnson2@nih.gov.
Organizational Affiliation: