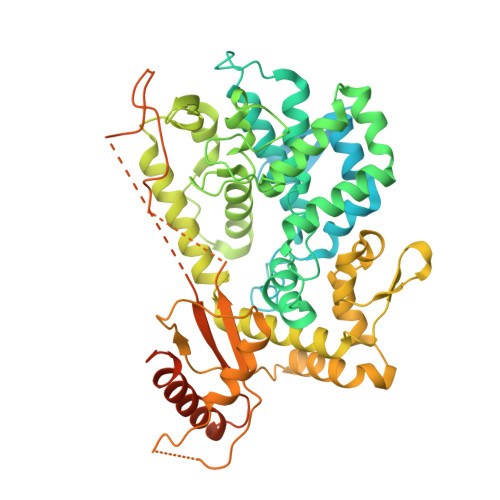
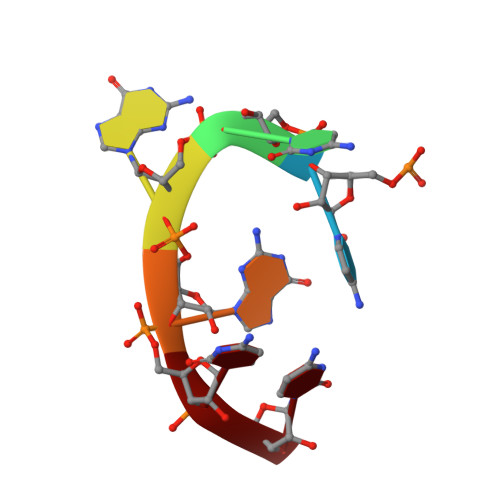
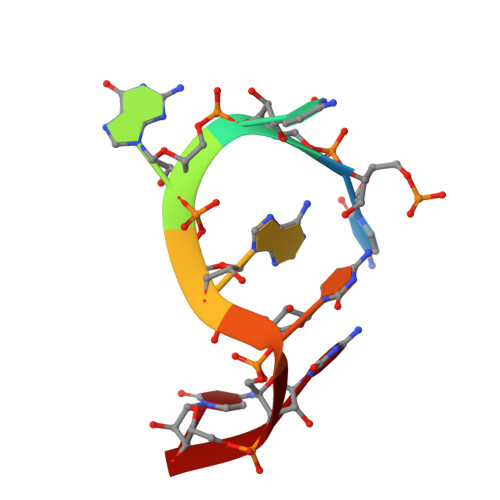
Guanine-containing ssDNA and RNA induce dimeric and tetrameric structural forms of SAMHD1.
Orris, B., Sung, M.W., Bhat, S., Xu, Y., Huynh, K.W., Han, S., Johnson, D.C., Bosbach, B., Shields, D.J., Stivers, J.T.(2023) Nucleic Acids Res 51: 12443-12458
- PubMed: 37930833 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gkad971
- Primary Citation Related Structures:
8TDV, 8TDW - PubMed Abstract:
The dNTPase activity of tetrameric SAM and HD domain containing deoxynucleoside triphosphate triphosphohydrolase 1 (SAMHD1) plays a critical role in cellular dNTP regulation. SAMHD1 also associates with stalled DNA replication forks, DNA repair foci, ssRNA and telomeres. The above functions require nucleic acid binding by SAMHD1, which may be modulated by its oligomeric state. Here we establish in cryo-EM and biochemical studies that the guanine-specific A1 activator site of each SAMHD1 monomer is used to target the enzyme to guanine nucleotides within single-stranded (ss) DNA and RNA. Remarkably, nucleic acid strands containing a single guanine base induce dimeric SAMHD1, while two or more guanines with ∼20 nucleotide spacing induce a tetrameric form. A cryo-EM structure of ssRNA-bound tetrameric SAMHD1 shows how ssRNA strands bridge two SAMHD1 dimers and stabilize the structure. This ssRNA-bound tetramer is inactive with respect to dNTPase and RNase activity.
- Department of Pharmacology and Molecular Sciences, Johns Hopkins University School of Medicine 725 North Wolfe Street Baltimore, MD 21205, USA.
Organizational Affiliation: