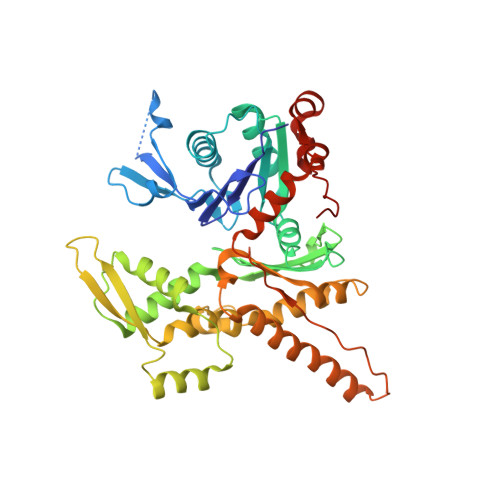
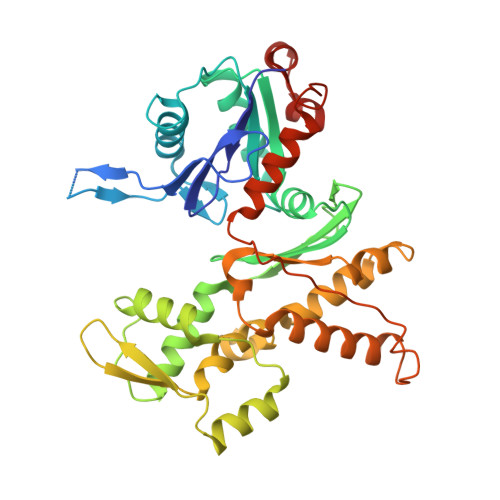
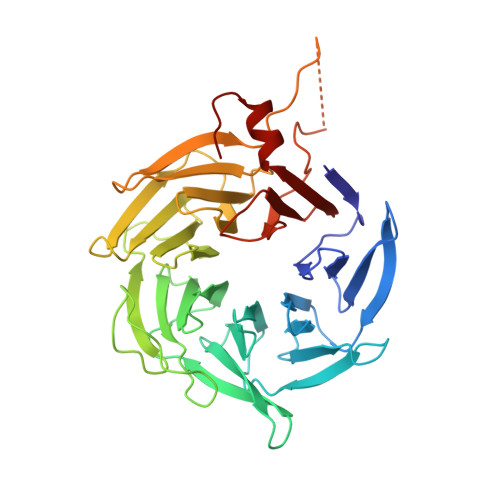
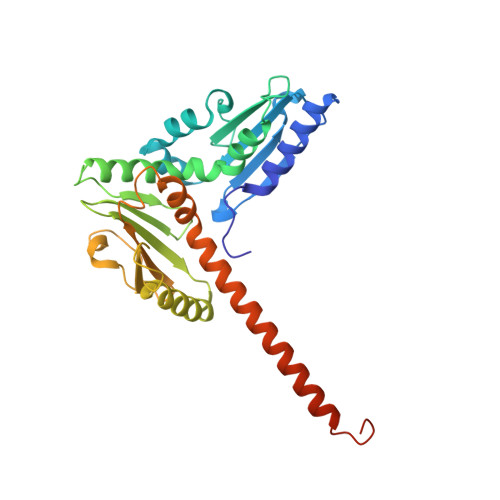
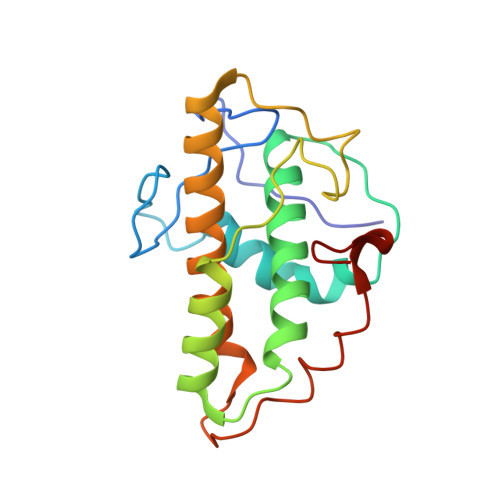
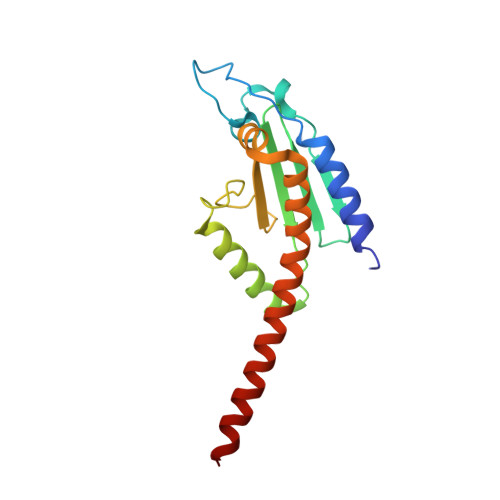
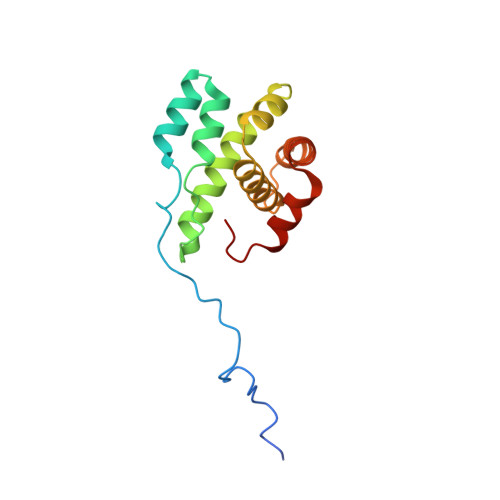
Mechanism of synergistic activation of Arp2/3 complex by cortactin and WASP-family proteins.
Fregoso, F.E., Boczkowska, M., Rebowski, G., Carman, P.J., van Eeuwen, T., Dominguez, R.(2023) Nat Commun 14: 6894-6894
- PubMed: 37898612 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-023-42229-y
- Primary Citation Related Structures:
8TAH - PubMed Abstract:
Cortactin coactivates Arp2/3 complex synergistically with WASP-family nucleation-promoting factors (NPFs) and stabilizes branched networks by linking Arp2/3 complex to F-actin. It is poorly understood how cortactin performs these functions. We describe the 2.89 Å resolution cryo-EM structure of cortactin's N-terminal domain (Cort 1-76 ) bound to Arp2/3 complex. Cortactin binds Arp2/3 complex through an inverted Acidic domain (D20-V29), which targets the same site on Arp3 as the Acidic domain of NPFs but with opposite polarity. Sequences N- and C-terminal to cortactin's Acidic domain do not increase its affinity for Arp2/3 complex but contribute toward coactivation with NPFs. Coactivation further increases with NPF dimerization and for longer cortactin constructs with stronger binding to F-actin. The results suggest that cortactin contributes to Arp2/3 complex coactivation with NPFs in two ways, by helping recruit the complex to F-actin and by stabilizing the short-pitch (active) conformation, which are both byproducts of cortactin's core function in branch stabilization.
- Department of Physiology, Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA, USA.
Organizational Affiliation: