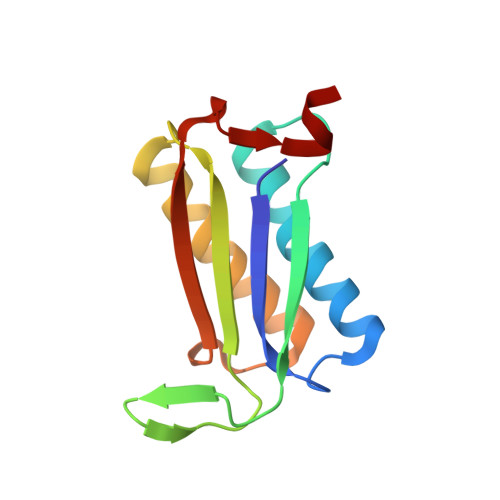
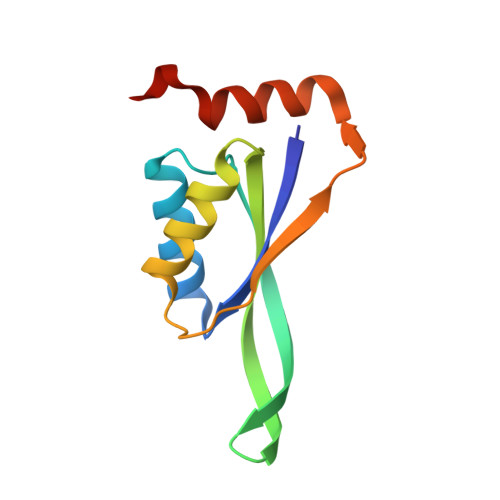
Rapid and automated design of two-component protein nanomaterials using ProteinMPNN.
de Haas, R.J., Brunette, N., Goodson, A., Dauparas, J., Yi, S.Y., Yang, E.C., Dowling, Q., Nguyen, H., Kang, A., Bera, A.K., Sankaran, B., de Vries, R., Baker, D., King, N.P.(2024) Proc Natl Acad Sci U S A 121: e2314646121-e2314646121
- PubMed: 38502697 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.2314646121
- Primary Citation Related Structures:
8T6C, 8T6E, 8T6N - PubMed Abstract:
The design of protein-protein interfaces using physics-based design methods such as Rosetta requires substantial computational resources and manual refinement by expert structural biologists. Deep learning methods promise to simplify protein-protein interface design and enable its application to a wide variety of problems by researchers from various scientific disciplines. Here, we test the ability of a deep learning method for protein sequence design, ProteinMPNN, to design two-component tetrahedral protein nanomaterials and benchmark its performance against Rosetta. ProteinMPNN had a similar success rate to Rosetta, yielding 13 new experimentally confirmed assemblies, but required orders of magnitude less computation and no manual refinement. The interfaces designed by ProteinMPNN were substantially more polar than those designed by Rosetta, which facilitated in vitro assembly of the designed nanomaterials from independently purified components. Crystal structures of several of the assemblies confirmed the accuracy of the design method at high resolution. Our results showcase the potential of deep learning-based methods to unlock the widespread application of designed protein-protein interfaces and self-assembling protein nanomaterials in biotechnology.
- Department of Physical Chemistry and Soft Matter, Wageningen University and Research, Wageningen 6078 WE, The Netherlands.
Organizational Affiliation: