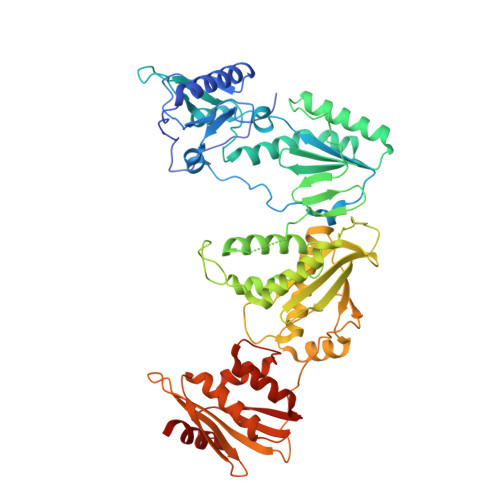
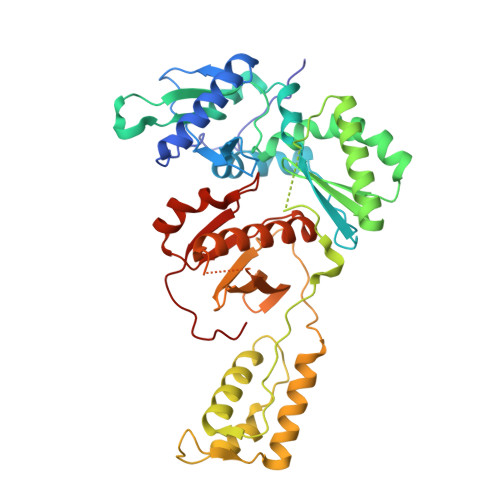
Exploring novel HIV-1 reverse transcriptase inhibitors with drug-resistant mutants: A double mutant surprise.
Hollander, K., Chan, A.H., Frey, K.M., Hunker, O., Ippolito, J.A., Spasov, K.A., Yeh, Y.J., Jorgensen, W.L., Ho, Y.C., Anderson, K.S.(2023) Protein Sci 32: e4814-e4814
- PubMed: 37861472
- DOI: https://doi.org/10.1002/pro.4814
- Primary Citation of Related Structures:
8STP, 8STQ, 8STR, 8STS, 8STT, 8STU, 8STV - PubMed Abstract:
HIV-1 reverse transcriptase (RT) remains a key target for HIV drug development. As successful management of the disease requires lifelong treatment, the emergence of resistance mutations is inevitable, making development of new RT inhibitors, which remain effective against resistant variants crucial. To this end, previous computationally guided drug design efforts have resulted in catechol diether compounds, which inhibit wildtype RT with picomolar affinities and appear to be promising preclinical candidates. To confirm that these compounds remain potent against Y181C, a widespread mutation conferring resistance to first generation inhibitors, they were screened against the HIV-1 N119 clinical isolate, reported as a Y181C single mutant. In comparison to a molecular clone with the same mutation, N119 appears less susceptible to inhibition by our preclinical candidate compounds. A more detailed sequencing effort determined that N119 was misidentified and carries V106A in combination with Y181C. While both indolizine and naphthalene substituted catechol diethers are potent against the classical Y181C single mutant, the addition of V106A confers more resistance against the indolizine derivatives than the naphthalene derivatives. Crystal structures presented in this study highlight key features of the naphthyl group, which allow these compounds to remain potent in the double mutant, including stronger interactions with F227 and less reliance on V106 for stabilization of the ethoxy-uracil ring, which makes critical hydrogen bonds with other residues in the binding pocket.
- Department of Molecular Biophysics and Biochemistry, Yale University School of Medicine, New Haven, Connecticut, USA.
Organizational Affiliation: