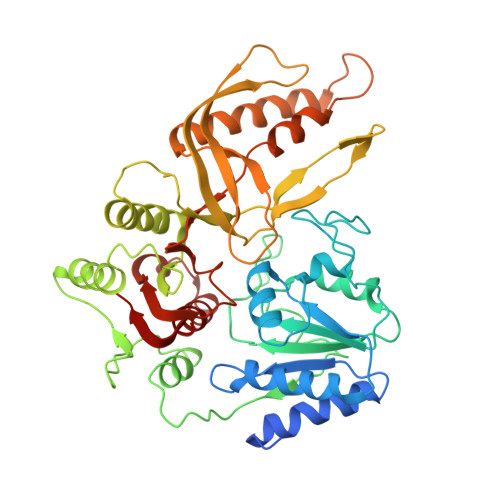
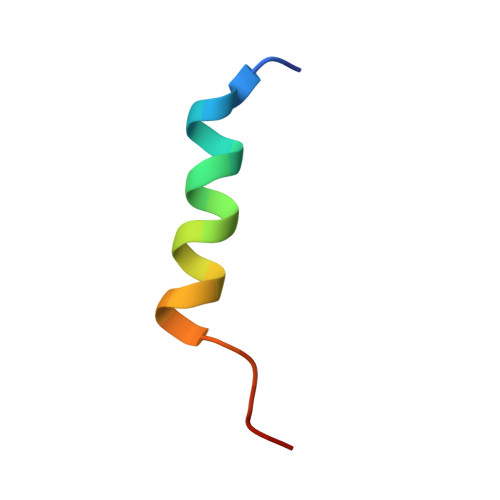
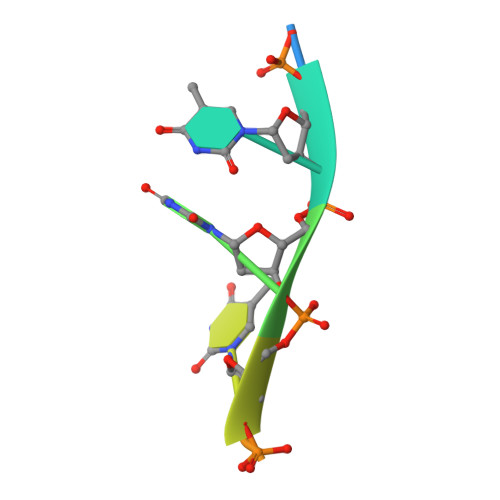
Structural and functional insights into the interaction between the bacteriophage T4 DNA processing proteins gp32 and Dda.
He, X., Yun, M.K., Li, Z., Waddell, M.B., Nourse, A., Churion, K.A., Kreuzer, K.N., Byrd, A.K., White, S.W.(2024) Nucleic Acids Res 52: 12748-12762
- PubMed: 39417586 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gkae910
- Primary Citation Related Structures:
8GME, 8S9I - PubMed Abstract:
Bacteriophage T4 is a classic model system for studying the mechanisms of DNA processing. A key protein in T4 DNA processing is the gp32 single-stranded DNA-binding protein. gp32 has two key functions: it binds cooperatively to single-stranded DNA (ssDNA) to protect it from nucleases and remove regions of secondary structure, and it recruits proteins to initiate DNA processes including replication and repair. Dda is a T4 helicase recruited by gp32, and we purified and crystallized a gp32-Dda-ssDNA complex. The low-resolution structure revealed how the C-terminus of gp32 engages Dda. Analytical ultracentrifugation analyses were consistent with the crystal structure. An optimal Dda binding peptide from the gp32 C-terminus was identified using surface plasmon resonance. The crystal structure of the Dda-peptide complex was consistent with the corresponding interaction in the gp32-Dda-ssDNA structure. A Dda-dependent DNA unwinding assay supported the structural conclusions and confirmed that the bound gp32 sequesters the ssDNA generated by Dda. The structure of the gp32-Dda-ssDNA complex, together with the known structure of the gp32 body, reveals the entire ssDNA binding surface of gp32. gp32-Dda-ssDNA complexes in the crystal are connected by the N-terminal region of one gp32 binding to an adjacent gp32, and this provides key insights into this interaction.
- Department of Structural Biology, St. Jude Children's Research Hospital, 262 Danny Thomas Place MS311, Memphis, TN 38105, USA.
Organizational Affiliation: