RAIN: a Machine Learning-based identification for HIV-1 bNAbs.
Perez, L., Foglierini, M.(2024) Res Sq
- PubMed: 38903123 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.21203/rs.3.rs-4023897/v1
- Primary Citation Related Structures:
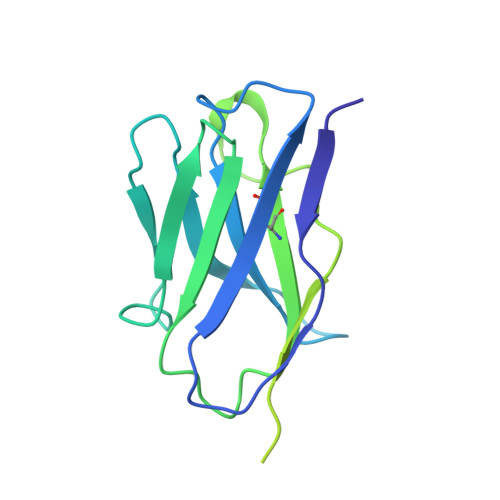
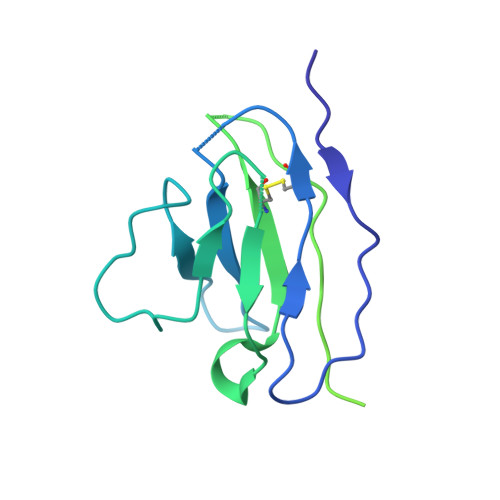
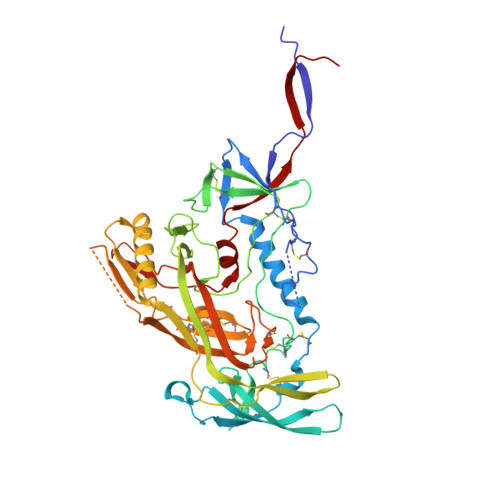
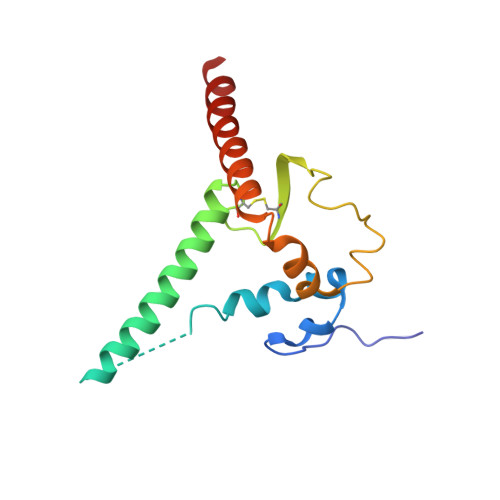
8S2E - PubMed Abstract:
Broadly neutralizing antibodies (bNAbs) are promising candidates for the treatment and prevention of HIV-1 infection. Despite their critical importance, automatic detection of HIV-1 bNAbs from immune repertoire is still lacking. Here, we developed a straightforward computational method for R apid A utomatic I dentification of b N Abs (RAIN ) based on Machine Learning methods. In contrast to other approaches using one-hot encoding amino acid sequences or structural alignment for prediction, RAIN uses a combination of selected sequence-based features for accurate prediction of HIV-1 bNAbs. We demonstrate the performance of our approach on non-biased, experimentally obtained sequenced BCR repertoires from HIV-1 immune donors. RAIN processing leads to the successful identification of novel HIV-1 bNAbs targeting the CD4-binding site of the envelope glycoprotein. In addition, we validate the identified bNAbs using in vitro neutralization assay and we solve the structure of one of them in complex with the soluble native-like heterotrimeric envelope glycoprotein by single-particle cryo-electron microscopy (cryo-EM). Overall, we propose a method to facilitate and accelerate HIV-1 bNAbs discovery from non-selected immune repertoires.
- Lausanne University Hospital and University of Lausanne, Lausanne, Switzerland.
Organizational Affiliation: