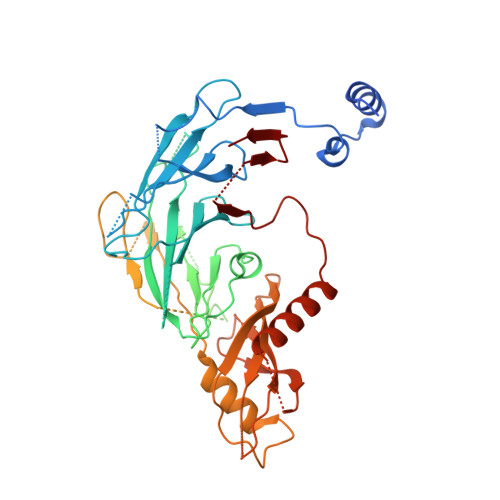
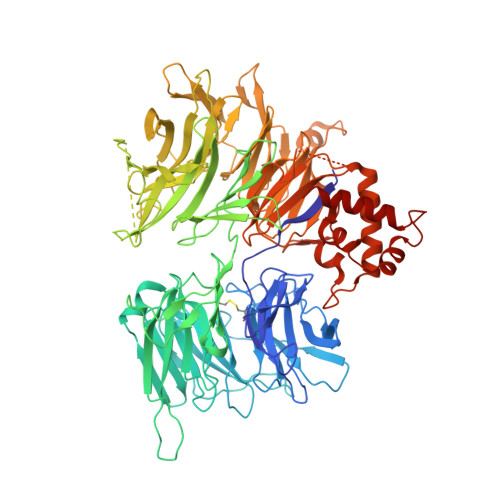
Optimization of Potent Ligands for the E3 Ligase DCAF15 and Evaluation of Their Use in Heterobifunctional Degraders.
Lucas, S.C.C., Ahmed, A., Ashraf, S.N., Argyrou, A., Bauer, M.R., De Donatis, G.M., Demanze, S., Eisele, F., Fusani, L., Hock, A., Kadamur, G., Li, S., Macmillan-Jones, A., Michaelides, I.N., Phillips, C., Rehnstrom, M., Richter, M., Rodrigo-Brenni, M.C., Shilliday, F., Wang, P., Storer, R.I.(2024) J Med Chem 67: 5538-5566
- PubMed: 38513086 Search on PubMed
- DOI: https://doi.org/10.1021/acs.jmedchem.3c02136
- Primary Citation Related Structures:
8ROX, 8ROY - PubMed Abstract:
Unlocking novel E3 ligases for use in heterobifunctional PROTAC degraders is of high importance to the pharmaceutical industry. Over-reliance on the current suite of ligands used to recruit E3 ligases could limit the potential of their application. To address this, potent ligands for DCAF15 were optimized using cryo-EM supported, structure-based design to improve on micromolar starting points. A potent binder, compound 24 , was identified and subsequently conjugated into PROTACs against multiple targets. Following attempts on degrading a number of proteins using DCAF15 recruiting PROTACs, only degradation of BRD4 was observed. Deconvolution of the mechanism of action showed that this degradation was not mediated by DCAF15, thereby highlighting both the challenges faced when trying to expand the toolbox of validated E3 ligase ligands for use in PROTAC degraders and the pitfalls of using BRD4 as a model substrate.
- Hit Discovery, Discovery Sciences, R&D, AstraZeneca, Cambridge CB2 0AA, U.K.
Organizational Affiliation: