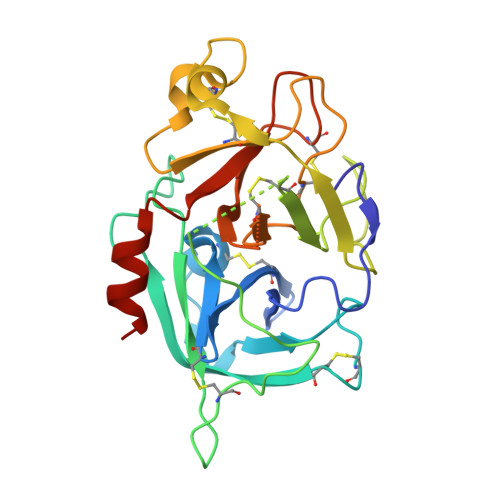
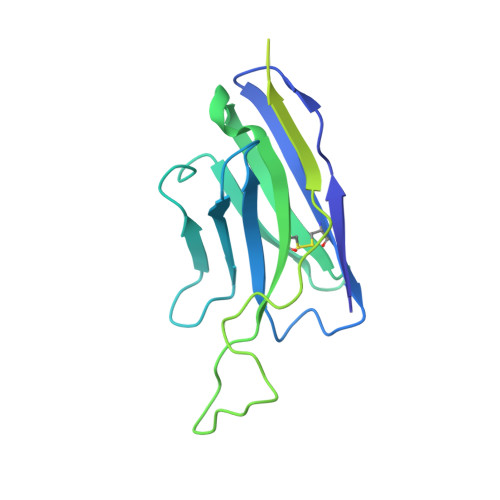
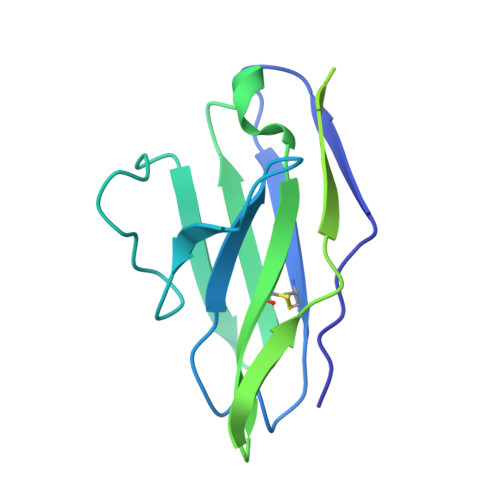
Structural basis for the inhibition of beta FXIIa by garadacimab.
Drulyte, I., Ghai, R., Ow, S.Y., Kapp, E.A., Quek, A.J., Panousis, C., Wilson, M.J., Nash, A.D., Pelzing, M.(2024) Structure 32: 1705
- PubMed: 39059382 Search on PubMed
- DOI: https://doi.org/10.1016/j.str.2024.07.001
- Primary Citation Related Structures:
8R8D - PubMed Abstract:
Activated FXII (FXIIa) is the principal initiator of the plasma contact system and can activate both procoagulant and proinflammatory pathways. Its activity is important in the pathophysiology of hereditary angioedema (HAE). Here, we describe a high-resolution cryoelectron microscopy (cryo-EM) structure of the beta-chain from FXIIa (βFXIIa) complexed with the Fab fragment of garadacimab. Garadacimab binds to βFXIIa through an unusually long CDR-H3 that inserts into the S1 pocket in a non-canonical way. This structural mechanism is likely the primary contributor to the inhibition of activated FXIIa proteolytic activity in HAE. Garadacimab Fab-βFXIIa structure also reveals critical determinants of high-affinity binding of garadacimab to activated FXIIa. Structural analysis with other bona fide FXIIa inhibitors, such as benzamidine and C1-INH, reveals a surprisingly similar mechanism of βFXIIa inhibition by garadacimab. In summary, the garadacimab Fab-βFXIIa structure provides crucial insights into its mechanism of action and delineates primary and auxiliary paratopes/epitopes.
- Materials and Structural Analysis, Thermo Fisher Scientific, Eindhoven, the Netherlands.
Organizational Affiliation: