Structure of human butyrylcholinesterase with (3-(((2-cycloheptylethyl)(methyl)amino)methyl)-1H-indol-7-yl)(methyl)carbamoylated Ser198
Brazzolotto, X., Meden, A., Knez, D., Gobec, S., Nachon, F.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cholinesterase | 529 | Homo sapiens | Mutation(s): 4 Gene Names: BCHE, CHE1 EC: 3.1.1.8 | 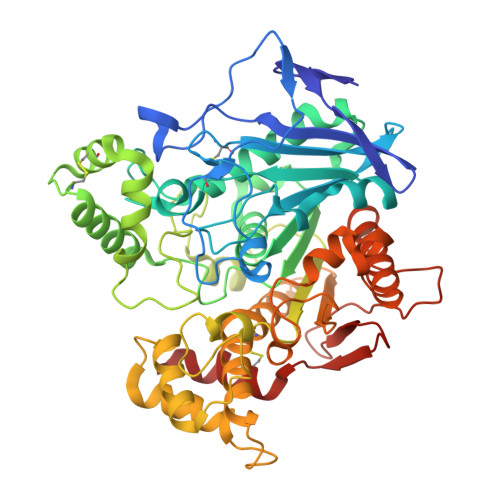 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P06276 GTEx: ENSG00000114200 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P06276 | ||||
Glycosylation | |||||
| Glycosylation Sites: 6 | Go to GlyGen: P06276-1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| alpha-L-fucopyranose-(1-6)-2-acetamido-2-deoxy-beta-D-glucopyranose | B, F | 2 |  | N-Glycosylation | |
Glycosylation Resources | |||||
GlyTouCan: G86851RC GlyCosmos: G86851RC GlyGen: G86851RC | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-[alpha-L-fucopyranose-(1-6)]2-acetamido-2-deoxy-beta-D-glucopyranose | C | 4 |  | N-Glycosylation | |
Glycosylation Resources | |||||
GlyTouCan: G32152BH GlyCosmos: G32152BH GlyGen: G32152BH | |||||
| Ligands 7 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| WXR (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | O [auth A] | N-[3-[[2-cycloheptylethyl(methyl)amino]methyl]-1H-indol-7-yl]-N-methyl-methanamide C21 H31 N3 O QISLTHXRTOUKIF-UHFFFAOYSA-N |  | ||
| SIA Download:Ideal Coordinates CCD File | N [auth A] | N-acetyl-alpha-neuraminic acid C11 H19 N O9 SQVRNKJHWKZAKO-YRMXFSIDSA-N |  | ||
| NAG Download:Ideal Coordinates CCD File | G [auth A] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| MES Download:Ideal Coordinates CCD File | J [auth A] | 2-(N-MORPHOLINO)-ETHANESULFONIC ACID C6 H13 N O4 S SXGZJKUKBWWHRA-UHFFFAOYSA-N |  | ||
| SO4 Download:Ideal Coordinates CCD File | Q [auth A], R [auth A], S [auth A], T [auth A] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| GOL Download:Ideal Coordinates CCD File | H [auth A], I [auth A], K [auth A], M [auth A], P [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| GOA Download:Ideal Coordinates CCD File | L [auth A] | GLYCOLIC ACID C2 H4 O3 AEMRFAOFKBGASW-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 154.231 | α = 90 |
| b = 154.231 | β = 90 |
| c = 127.979 | γ = 90 |
| Software Name | Purpose |
|---|---|
| MxCuBE | data collection |
| autoPROC | data processing |
| XDS | data reduction |
| XSCALE | data scaling |
| PHASER | phasing |
| Coot | model building |
| PHENIX | refinement |
| Funding Organization | Location | Grant Number |
|---|---|---|
| French Ministry of Armed Forces | France | NBC-5-C-2316 |