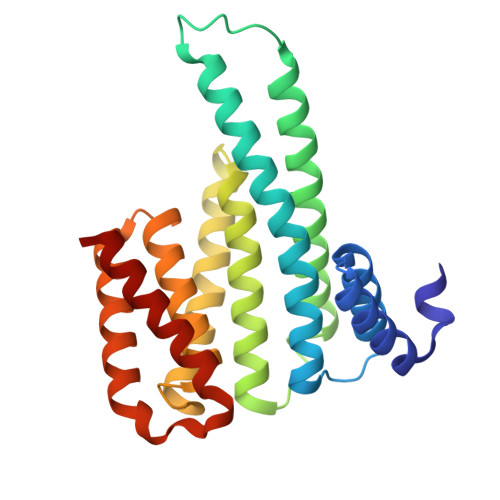
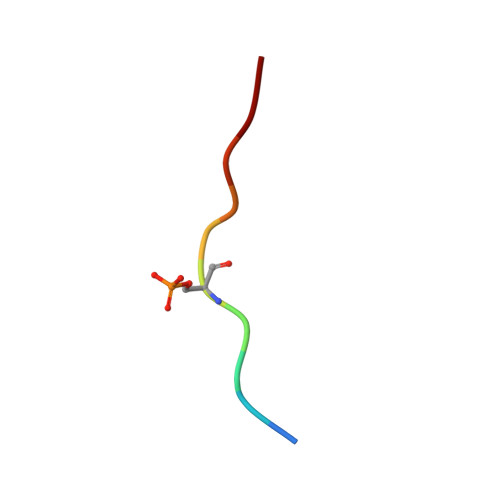
Modulation of the 14-3-3 sigma /C-RAF "Auto"inhibited Complex by Molecular Glues.
Konstantinidou, M., Vickery, H.R., Pennings, M.A.M., Virta, J.M., Luo, S.Y., Visser, E.J., Bannier, S.D., Srikanth, M., Cismoski, S.Z., Young, L.C., van den Oetelaar, M.C.M., McCormick, F., Ottmann, C., Brunsveld, L., Arkin, M.R.(2026) J Am Chem Soc 148: 4951-4965
- PubMed: 41617210 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/jacs.5c12622
- Primary Citation Related Structures:
8Q55, 8Q5C, 8QS2, 8QS3, 8QS4, 8QS5, 8QS6, 8QS7, 8QS8, 8QS9, 8QSA, 8QSB, 8QSC, 8QSD, 8QSE, 8QSF, 8QSG, 8QSH, 8S42, 9EW1, 9EW3, 9EW4, 9EW5, 9EW6, 9EW7 - PubMed Abstract:
Molecular glues, compounds that bind cooperatively at protein-protein interfaces (PPIs), are revolutionizing chemical biology and drug discovery, allowing the modulation of traditional "undruggable" targets. Here, we focus on a native regulatory PPI between the scaffolding protein 14-3-3 and C-RAF, a key component of the MAPK signaling pathway. Extensive drug discovery efforts have focused on the MAPK pathway due to its central role in oncology and developmental disorders (RASopathies). However, the modulation of its protein complexes is underexplored. C-RAF activity is regulated on multiple levels including dimerization, phosphorylation, and complex formation with 14-3-3, which prevents C-RAF activation by binding to a C-RAF sequence centered on phospho-serine 259. We used a fragment-merging approach to design molecular glues that bound to the composite surface of this 14-3-3/C-RAFpS259 complex. Molecular glues stabilized the inhibitory complex up to 300-fold; their glue-based mechanism of action was confirmed by crystallography and biophysical studies. Selectivity among the other RAF isoforms and other RAF phosphorylation sites was evaluated. The best compounds showed excellent selectivity among a broad panel of 80 14-3-3 clients. Cellular assays demonstrated on-target engagement, enhanced phosphorylation levels of C-RAFpS259, and reduced levels of RAF dimerization and ERK phosphorylation. Overall, this approach enabled chemical biology studies for a C-RAF site that was intrinsically disordered prior to 14-3-3 binding and had not been targeted previously. These molecular glues will be useful chemical probes and starting points for drug discovery efforts to modulate native PPI stabilization in the MAPK pathway with applications in oncology and RASopathies.
- Department of Pharmaceutical Chemistry and Small Molecule Discovery Center (SMDC), University of California San Francisco, San Francisco, California 94143, United States.
Organizational Affiliation: