Discovery and Exploration of Protein Kinase CK2 Binding Sites Using CK2alpha Cys336Ser as an Exquisite Crystallographic Tool
Werner, C., Lindenblatt, D., Viht, K., Uri, A., Niefind, K.(2023) Kinases Phosphatases
Experimental Data Snapshot
Starting Model: experimental
View more details
(2023) Kinases Phosphatases
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Casein kinase II subunit alpha' | 364 | Homo sapiens | Mutation(s): 1 Gene Names: CSNK2A2, CK2A2 EC: 2.7.11.1 | 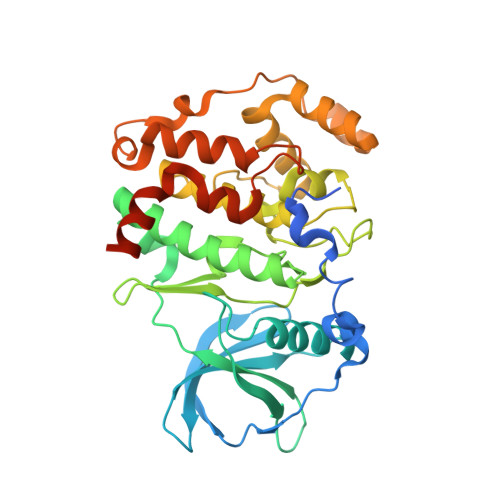 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P19784 GTEx: ENSG00000070770 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P19784 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| LN3 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | B [auth A], C [auth A] | (2~{S})-2-[[(2~{S})-2-[[(2~{S})-2-[12-[[4-[[5-(4-carboxyphenyl)-1,3-thiazol-2-yl]amino]-4-oxidanylidene-butanoyl]-(2-hydroxy-2-oxoethyl)amino]dodecanoylamino]-4-oxidanyl-4-oxidanylidene-butanoyl]amino]-4-oxidanyl-4-oxidanylidene-butanoyl]amino]butanedioic acid C40 H52 N6 O17 S GSTABKNLIUJVNF-QKDODKLFSA-N |  | ||
| EDO Download:Ideal Coordinates CCD File | D [auth A] E [auth A] F [auth A] G [auth A] H [auth A] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| CL Download:Ideal Coordinates CCD File | J [auth A] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 46.603 | α = 66.59 |
| b = 47.919 | β = 89.67 |
| c = 51.031 | γ = 88.16 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XDS | data reduction |
| Aimless | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| German Research Foundation (DFG) | Germany | NI 643/11-1 |
| German Research Foundation (DFG) | Germany | NI 643/4-2 |