Crystal structure of UDP/mannose-bound AGO61/beta-1,4-N-Acetylglucosaminyltransferase 2 (POMGNT2)
Satoh, T., Umezawa, F., Yagi, H., Kato, K.To be published.
Experimental Data Snapshot
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Protein O-linked-mannose beta-1,4-N-acetylglucosaminyltransferase 2 | 558 | Homo sapiens | Mutation(s): 0 Gene Names: POMGNT2, AGO61, C3orf39, EOGTL, GTDC2 EC: 2.4.1.312 | 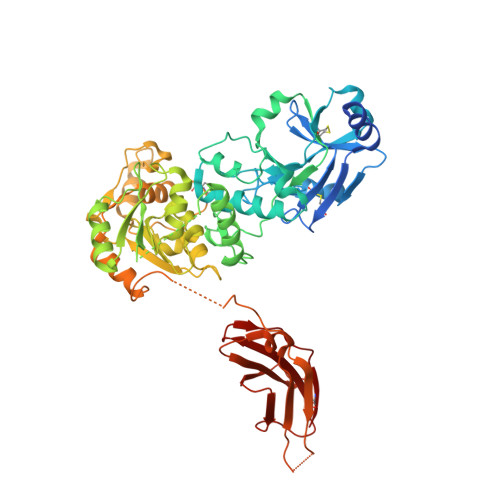 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q8NAT1 GTEx: ENSG00000144647 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q8NAT1 | ||||
Glycosylation | |||||
| Glycosylation Sites: 1 | Go to GlyGen: Q8NAT1-1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| UDP (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | E [auth A], J [auth B], N [auth C], S [auth D] | URIDINE-5'-DIPHOSPHATE C9 H14 N2 O12 P2 XCCTYIAWTASOJW-XVFCMESISA-N |  | ||
| NAG Download:Ideal Coordinates CCD File | F [auth A] G [auth A] K [auth B] O [auth C] P [auth C] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| MAN (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | H [auth A], L [auth B], Q [auth C], U [auth D] | alpha-D-mannopyranose C6 H12 O6 WQZGKKKJIJFFOK-PQMKYFCFSA-N |  | ||
| CL Download:Ideal Coordinates CCD File | I [auth A], M [auth B], R [auth C], V [auth D] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 145.019 | α = 90 |
| b = 150.002 | β = 90 |
| c = 191.481 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| XDS | data reduction |
| XDS | data scaling |
| AutoSol | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Japan Agency for Medical Research and Development (AMED) | Japan | 23gm6410010h0004 |
| Ministry of Education, Culture, Sports, Science and Technology (Japan) | Japan | JP19H03361 |